
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.4, Problem 2.7P
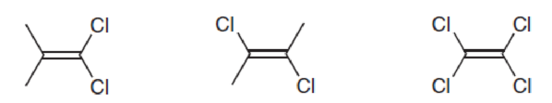
Predict which of the following
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Predict the theoretical number of different NMR signals produced by each compound, and give approximate chemical shifts. Point out any diastereotopic relationships.
(a) Ph¬CHBr¬CH2Br (b) vinyl chloride
4 Which of these C6H14 isomers has the greatest number of ¹3C NMR signals? (a) Hexane (d) 2,2- (b) Dimethylbutane 2-Methylpentane X3-Methylpentane (e) 2.3- Dimethylbutane
Splitting patterns in proton NMR spectra convey information on a hydrogen’s neighboring positions. A signal is split into n+1 number of peaks where the “n” is the number of protons on adjacent carbons. Indicate how many peaks you would expect to see in the signal of the indicated hydrogens.
Chapter 2 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - The following compound has three carbonyl groups....Ch. 2.4 - Predict which of the following C=C bonds will...Ch. 2.4 - The C=C bond in the following compound produces an...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...
Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.6 - Prob. 2.22P
Additional Science Textbook Solutions
Find more solutions based on key concepts
A compound that contains only C and H was burned in excess O2 to give CO2 and H2O. When 0.270 g of the compound...
General Chemistry: Atoms First
48. Are these data sets on chemical changes consistent with the law of conservation of mass?
a. A 12.8-g sample...
Introductory Chemistry (5th Edition) (Standalone Book)
A flat plate is of planar dimension 1m0.75m. For parallel laminar flow over the plate, calculate the ratio of t...
Fundamentals of Heat and Mass Transfer
9.1 Calculate the total mass of the reactants and the products for each of the following equations:
Basic Chemistry (5th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
What is the wavelength, in nanometers, of light with an energy content of 2112 kJ/mol? In what portion of the e...
General Chemistry: Principles and Modern Applications (11th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the molecules below would produce the following H-NMR spectrum? Select one: 1. A 2. D 3. B 4. E 5. Carrow_forwardNMR. Indicate how many signals appear in the proton spectrum of the(1) CICH=CH2(2) o-chloromethylbenzenearrow_forwardTrue or False 1. A molecule that is "IR inactive" means that it does not produce any signal due to no vibration. 2. Infrared spectroscopic data is reported in wavenumber (cm-1) against absorbance because they have a linear relationship. 3. The signals observed from a molecule of chloropropane will have a higher wavenumber than iodopropane. 4. The signals observed from the C-C bond in an alkene will report at a higher wavenumber than the C-C bond in an alkyne.arrow_forward
- Draw the 1H spectrum of the following compoundarrow_forwardConsidering IR spectroscopy, which of the following statements is incorrect? options: Concentrated alcohols give rise to broad signals while dilute alcohols give rise to narrow signals Carboxylic acids exist as dimer due to hydrogen bonding and thus produce a narrow signal at 3600 cm-1 and a characteristic signal due to C=O at 1700 cm-1 C=O bonds produce strong signals in an IR spectrum while C=C bonds often produce a weak signals Symmetrical C=C bonds and C≡C bonds do not produce signals Primary amines produce two signals resulting from symmetric stretching and asymmetric stretchingarrow_forwardBelow are three MS spectra, Spectra 1, 2, and 3. Each of this mass spectrum corresponds to either Compound A (contains one Cl in the molecular formula), Compound B (one I in the molecular formula), and Compound C (one Br in the molecular formula). Identify which spectra corresponds to which compound. Provide explanations for your choices.arrow_forward
- For a mono-substituted phenyl ring, which of the following absorbances would one expect to observe? Group of answer choices 880-900 730-770, 690-710 3000-3100 735-770arrow_forwardCarvone is an unsaturated ketone responsible for the odor of spearmint. If carvone has M+=150 in its mass spectrum and contains three double bonds and one ring, what is its molecular formula?arrow_forwardThe 1H and 13C NMR for a compound with the formula C5H12O are shown. Determine the structure of this compound. Assign the peaks: assign “a” to the most shielded hydrogen(s) in your structure, “b” to next-most shielded, etc; use capital letters for carbons).arrow_forward
- NMR: provide the structure of the following molecule. C5H10O- IR shows a strong, sharp signal at 1700arrow_forwardHow many peaks will show up in the H NMR spectrum of the given compound below. Choose one: A. 9 B. 6 C. 5 D. 11arrow_forwardC10H14. 500 MHZ. 1H NMR Can someone draw the structure showing all carbons and hydrogens and assign a pick value to all hydrogens and carbons from the spectra.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY