
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.5, Problem 2.20P
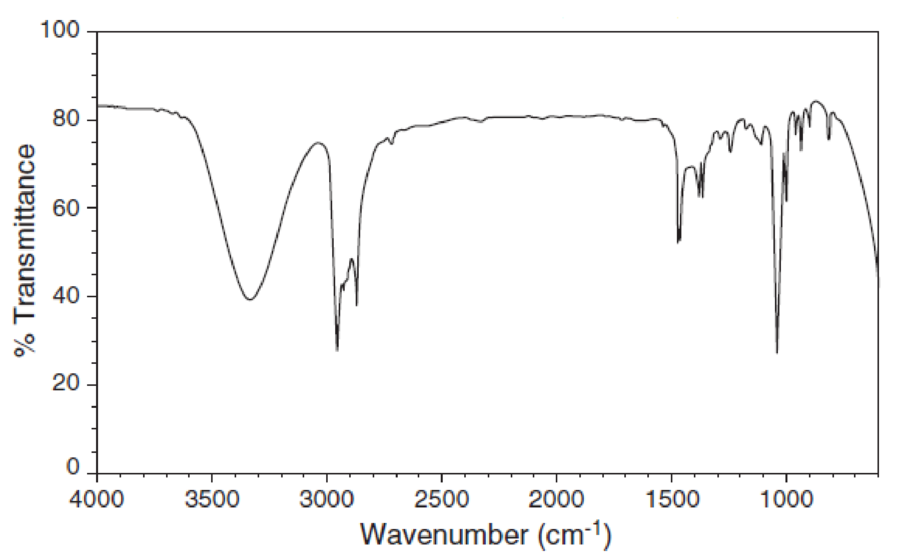
For each IR spectrum below, determine whether it is consistent with the structure of a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Based on this portion of an IR spectra is there a ketone that is present?
Draw the structure of C10H18O2 which is consistent with the 13C NMR data below. Thepeak appearing at 14 ppm is actually two very close peaks.
A student identified an organic unknown based on the IR spectrum as an aliphatic carboxylic acid. Explain what signal on a mass spectrum will also confirm that the unknown organic compound is a carboxylic acid.
Chapter 2 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - The following compound has three carbonyl groups....Ch. 2.4 - Predict which of the following C=C bonds will...Ch. 2.4 - The C=C bond in the following compound produces an...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...
Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.6 - Prob. 2.22P
Additional Science Textbook Solutions
Find more solutions based on key concepts
37. Balance each redox reaction occurring in acidic aqueous solution.
a. K(s) + Cr3+(aq) → Cr(s) + K+(aq)
b. Al...
Chemistry: A Molecular Approach
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
For each of the following, (i) give the systematic name of the compound and specify the oxidation state of the ...
General Chemistry: Atoms First
17.44 Cantharidin is a powerful vesicant that can be isolated from dried beetles (Cantharis vesicatoria, or “Sp...
Organic Chemistry
Practice Exercise 1.14
A crystal of salt was grown and found to have a mass of 0.547 g. When the crystal was pl...
Chemistry: The Molecular Nature of Matter
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A large doublet and a small septet pattern in 'H NMR is usually indicative of a(n) A. ethyl group.B. propyl group.C. isopropy1 group.D. phenyl group.arrow_forwardConsider carbons x, y, and z in p-methylanisole. One has a chemical shift of δ 20, another has δ 55, and the third δ 157. Match the chemical shifts with the appropriate carbons.arrow_forwardExplain why the gem-dimethyl groups appear as separate peaks in the proton-NMR spectrum of isoborneol, although they almost overlap in borneol.arrow_forward
- Consider isomeric alcohols A and B and mass spectra [1] and [2]. (a) Label the molecular ion and base peak in each spectrum. (b) Use thefragmentation patterns to determine which mass spectrum correspondsto isomer A and which corresponds to isomer B.arrow_forwardHow many (total) 13C NMR signals should be expected for this amine? b. Which one should be the farthest downfield (left) in the spectrum?arrow_forwardDetermine the structure from the FTIR and mass spectrum. The proton NMR has a singlet with an integration of 6 hydrogens at 2.6 ppm, a triplet with an integration of 1 hydrogen at 7.41 ppm, and a doublet with an integration of 3 hydrogens at 7.25 ppm.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY