
Concept explainers
(a)
Interpretation:
Given compound has to be prepared by Heck reaction using methyl-2-propenaote as the starting material.
Concept Introduction:
The
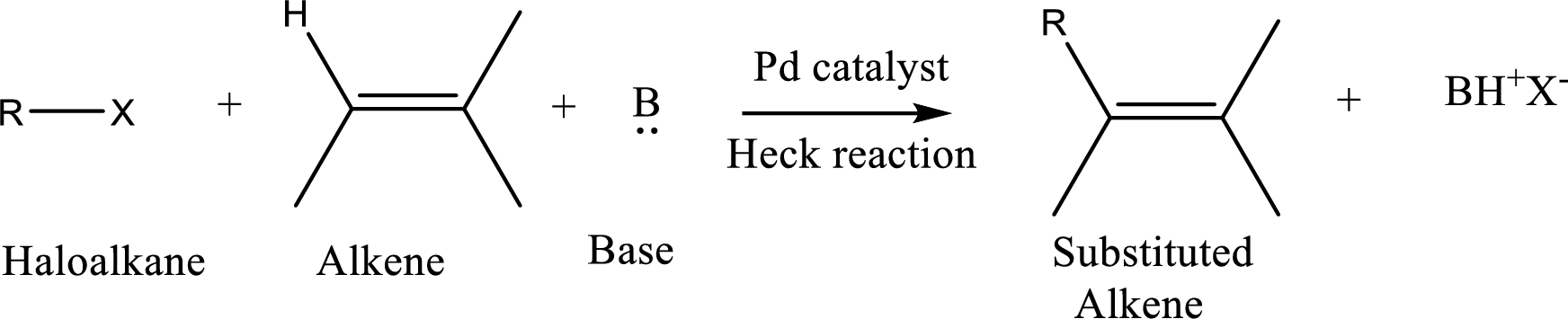
Reaction is highly regioselective in which formation of a new carbon-carbon bond occurs at a less substituted carbon of a double bond.
(b)
Interpretation:
Given compound has to be prepared by heck reaction using methyl-2-propenaote as the starting material.
Concept Introduction:
The
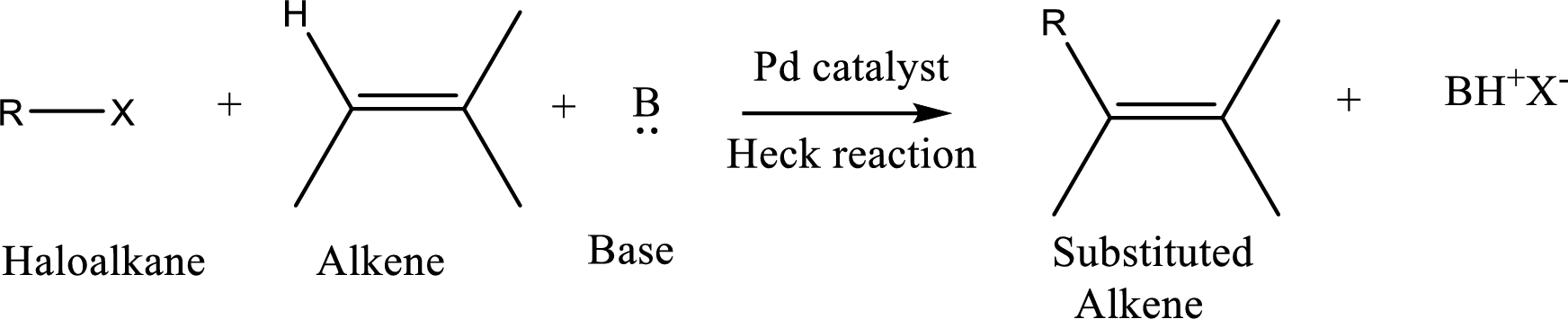
Reaction is highly regioselective in which formation of a new carbon-carbon bond occurs at a less substituted carbon of a double bond.
Trending nowThis is a popular solution!

Chapter 24 Solutions
Organic Chemistry
- Wolff-Kishner reduction of compound W gave compound A. Treatment of A with m-chloroperbenzoic acid gave B which on reduction with LiAH4 gave C. Oxidation of compound C with chromic acid gave D (C9H14O). Suggest the structures for A, B, C, and D.arrow_forwardGive the structure of compound 1 and compound 2arrow_forwardgive the structural formula for compounds A to Garrow_forward
- Compound A(C10H12O)gives off oxygen on treatment with sodium metal and also decolorizes Br2 in CCl4 to give organic compound B. Compound A on treatment with I2 in NaOH gives iodoform and salt C which after acidification gives a white solid D(C7H6O2). Using knowledge of organic chemistry identify structures A,B,C and Darrow_forwardGive the reagents and conditions necessary for the following conversion. A to B, B to D, B to C, B to E, E to F, E to G, G to H, H to I. Hence deduce the name and structural formula of the compounds C and I. Compare the procedure for converting F and E to G.arrow_forwardMention and Write the following reaction mechanisms: a) thermal dehydration of 4-t-butylcyclohexanol with phosphoric acid as catalyst; b) addition of molecular bromine to the C= C double bond of 4-t- butylcyclohexene;arrow_forward
- Offer at least two methods for the preparation: a) ethyl bromide, b) iso-propanol.arrow_forwardCompound A and compound B are in equilibrium. Write a stepwise mechanism from compound Ato compound B showing ALL intermediates. Use curved arrows to symbolize the flow of electrons to show how each of the intermediates and products are formed. Show all lone pairs and formal charges. Lastly, explain which compound (Aor B) will be in higher concentration.arrow_forwardPredict the reagents for reactions A and B.arrow_forward
- An organic compound A of unknown structure was found to have a molecular formula C8H16. When A was poured in water and heated, compound B having a molecular formula C8H18O was formed. B upon heating with sulfuric acid was converted to C as the major product which is identical to A. Ozonolysis of C gave one molecule each of two different products D and E, both having a molecular formula C4H8O. Write the reactions involved and determine the structure of A,B,C,D and E.arrow_forwardStarting with acetylene and ethylene oxide as the only sources of carbon atoms, show how to prepare the compound Q.)Hexanedialarrow_forwardDraw the structure of a compound of molecular formula C6H10 that reacts with H2 in the presence of Pd-C but does not react with H2 in the presence of Lindlar catalyst.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
