
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
4th Edition
ISBN: 9781119234722
Author: Klein
Publisher: JOHN WILEY+SONS,INC.-CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.5, Problem 2.14P
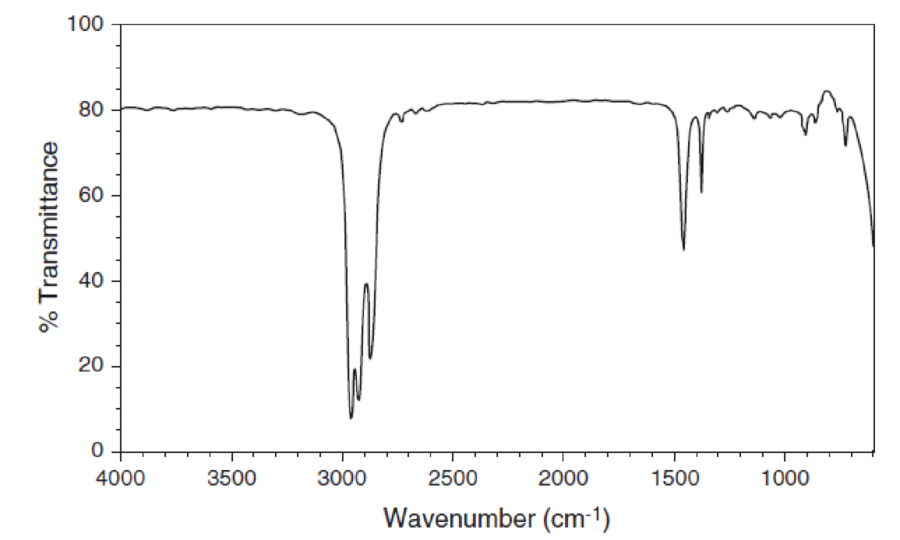
For each IR spectrum below, determine whether it is consistent with the structure of an alcohol, a
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Choose the molecule to which this IR spectrum corresponds.
5. Place the letter of the spectrum next to the name of the compound that it represents.
Compound
Letter
Acetone
1,2-dichloroethane
1,1,2-trichloroethane
2,2-dimethoxypropane
1-bromopropane
2-bromopropane
3. Which of the following information is primarily obtained from infrared spectroscopy?
A) arrangement of carbon and hydrogen atoms in a compound
B) molecular weight of a compound
C) any conjugated t system present in a compound
D) functional groups present in a compound
E) all of these
Chapter 2 Solutions
EBK ORGANIC CHEMISTRY AS A SECOND LANGU
Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - For each of the following compounds, determine...Ch. 2.3 - The following compound has three carbonyl groups....Ch. 2.4 - Predict which of the following C=C bonds will...Ch. 2.4 - The C=C bond in the following compound produces an...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...
Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, identify whether it is...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.5 - For each IR spectrum below, determine whether it...Ch. 2.6 - Prob. 2.22P
Additional Science Textbook Solutions
Find more solutions based on key concepts
9.1 Calculate the total mass of the reactants and the products for each of the following equations:
Basic Chemistry (5th Edition)
Draw structures for a saturated hydrocarbon that has a molecular ion with an m/z value of 128.
Organic Chemistry (8th Edition)
10.71 Identify each of the following as an acid or a base: (10.1)
H2SO4
RbOH
Ca(OH)2
HI
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Consider a cylindrical nuclear fuel rod of length L and diameter D that is encased in a concentric tube. Pressu...
Fundamentals of Heat and Mass Transfer
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
1. What did each of the following scientists contribute to our knowledge of the atom?
a. William Crookes
b. E...
Chemistry For Changing Times (14th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Each of the IR spectra below corresponds to one of the aromatic compounds listed below. Determine the molecule that corresponds to each spectrum.arrow_forwardThe DEPT-90 spectrum exhibits 6 in the 0-50 ppm region The DEPT-135 spectrum exhibits x 100 ppm region that is a positive ▾ C6 signal(s) for the CH groups: ▼ 1,2,6 ✓ in the sp2 hybridized region 100-150 C3 and C4 ▼ signal(s) (only the quaternary carbon atoms, signal(s), indicating the presence of a methylene group (CH₂) attached to an oxygen atom, are missing); there is C5 ▼ C1 and C2 ▼ and signal(s) in the 50-arrow_forwardTrue or False 1. A molecule that is "IR inactive" means that it does not produce any signal due to no vibration. 2. Infrared spectroscopic data is reported in wavenumber (cm-1) against absorbance because they have a linear relationship. 3. The signals observed from a molecule of chloropropane will have a higher wavenumber than iodopropane. 4. The signals observed from the C-C bond in an alkene will report at a higher wavenumber than the C-C bond in an alkyne.arrow_forward
- How do I interpret, label, and determine which molecule this H NMR graph represents? The three molecule choices are methylene cyclohexane, 3-methylcyclohexene, or 1-methylcyclohexene.arrow_forwardDetermine the product and other reactants based on the IR spectrum below.arrow_forwardAssign each peak on the following spectrum.arrow_forward
- 3. a) Draw the below structure and answer the following questions. CI CH2COCH2CH3 i. Identify the chemically equivalent protons in the structure. Explain your answer. ii. Mention how many peaks you would expect in the HNMR spectrum of the compound. Explain the position of each peak in the spectrum. iii. Mention what would be the splitting pattern of each peak with proper explanation. iv. Draw the HNMR spectrum of the compound. b) Explain why the delta/ ppm scale was introduced instead of Hz in NMR analysis with example.arrow_forwardWhat is this compound base on the IR spectrum?arrow_forwardspectrum shown on the following pages. Then please draw what you might expect their 'H spectra to look like. Most spectra in this problem set were obtained from the Spectral Database for Organic Compounds. HO, но HO,arrow_forward
- Below are three MS spectra, Spectra 1, 2, and 3. Each of this mass spectrum corresponds to either Compound A (contains one Cl in the molecular formula), Compound B (one I in the molecular formula), and Compound C (one Br in the molecular formula). Identify which spectra corresponds to which compound. Provide explanations for your choices.arrow_forward2a) Label the protons in each highlighted CH, group as homotopic, enantiotopic, or diastereotopic. Structure: Homotopic, Enantiotopic, or Diastereotopic? b) There is one structure in Question 1 that contains diastereotopic protons, leading to a higher number of signals than you originally may have expected. Circle the structure and revise (if necessary) the number of ¹H signals you expect that structure to exhibit.arrow_forwardWhich one of the pi bonds in the molecules shown below will produce a stronger signal in an IR spectrum? Yes, compound I produces a stronger signal because it has a larger dipole moment. O Yes, compound II produces a stronger signal because it has a larger dipole moment. No, the alkene groups produce similar signals because they have similar double bonds. O No, the alkene groups produce similar signals because they both contain a similar number of atoms.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY