
Principles of Instrumental Analysis, 6th Edition
6th Edition
ISBN: 9788131525579
Author: Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher: Cenage Learning
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 25, Problem 25.10QAP
Quinone undergoes a reversible reduction at a voltammetric working electrode. The reaction is
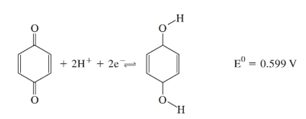
(a) Assume that the diffusion coefficients for quinone and hydroquinone are approximately the same and calculate the approximate half-wave Potential (versus SCE) for the reduction of hydroquinone at an RDE from a solution buffered to a pH of 8.0.
(b) Repeat the calculation in (a) for a solution buffered to a pH of 5.0.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Give the thermodynamic product for the following reaction
(b) Give the products for the following chemical reactions.
HO
SOCI,
CHO
NACN
(H)
(D
HCN
Suggest conditions for performing the following reaction:
Chapter 25 Solutions
Principles of Instrumental Analysis, 6th Edition
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the equilibrium-constant expressions and obtainnumerical values for each constant in. (a) the basic dissociation of aniline, C6H5NH2. (b) the acidic dissociation of hypochlorous acid,HClO. (c) the acidic dissociation of methyl ammoniumhydrochloride, CH3NH3Cl. (d) the basic dissociation of NaNO2. (e) the dissociation of H3AsO3to H3O+and AsO33-. (f) the reaction of C2O42-with H2O to give H2C2O4and OH-. show solutionarrow_forwardCyclopentadiene is especially acidic for a hydrocarbon. Use a diagram to explain why.arrow_forward4b please answer (predict the outcome and explain brieflyarrow_forward
- Redox Problem.Given reaction between dissolved Fe2+ and elemental sulfur (S0) to form hematite (Fe2O3) and sulfide HS- Fe2+ + S0 --> Fe2O3 + HS- (a) What the oxidant and reductant in this reaction? You must determine half-reactions and balance the reaction. (b) Is this reaction thermodynamically favorable? (c) Would this HS- to react readily with Fe2O3 (reverse of the reaction)?arrow_forwardAcidified potassium bromate(V), KBrO3, reacts with hydrogen sulfide, H2S, to give a yellow solid and an orange solution. On shaking the solution with trichloroethane, the trichloroethane layer turned orange-red. (a) Suggest the identity of yellow solid and the orange solution. Describe the type of reaction taking place. (b) Construct a balanced equation for the reaction between KBrO3 and H2S. (C) BrO3- is the conjugate base of HBrO3. Draw the dot and cross structure of HBrO3. (d) HBr is a stronger acid than HCl. However, HBrO3 is a weaker acid than HCIO3. Explain the difference in acidities.arrow_forward(d) onlyarrow_forward
- (b) Compare combustion of Anthracene and Xylene in sufficient supply of air. Which one will produce more carbon rich sooty flame and why?arrow_forwardK2Cr2O7 in acidic medium oxidises into which compound by H2O2 ?arrow_forward10. The following reaction has variation of free energy value of AG = -2.1 kJ/mol CH;Br + H2S 2 CH;SH + HBr (a) Calculate Keq at room temperature (25°C) for this reaction as written. (b) Starting with a 1 M solution of CH3B and H2S, calculate the final concentrations of all four species at equilibrium.arrow_forward
- S + Ozle) → SO26) 2 SO2e) + 02 ) → 2 SO31@) SO3e) + H2SO4laq) → H;S;O7(aq) H;S;O7(a) + H2O) → 2 H;SO(aq) (a) What is the oxidation number of S on the products of the first and second equations above? (b) What is being reduced in the second equation? (c) H2S,0, is known as oleum. What is the oxidation state of sulphur in this compound? (d) Is the fourth equation a redox equation? Explain your answer.arrow_forwardEthanoic acid and ethanol react together to form the ester ethyl ethanoate, CH₂COOC₂H, and water. CH₂COOH (1) + CH₂CH₂OH(I) = CH₂COOCH₂CH₂(1) + H₂O(l)arrow_forwardCalculate the standard free energy change (AG") for the transfer of electrons from succinate to ubiquinone: Succinate + ubiquinone O -2.7 kJ/mol relevant half-reactions and standard reduction potentials: fumarate + 2H* + 2 e™ H succinate ubiquinone + 2 H+ + 2 e² = O2.7 kJ/mol -8.7 kJ/mol O 14.7 kJ/mol succinate dehydrogenase O -6.0 kJ/mol Fumarate + ubiquinol ubiquinol E = 0.031 V ED - 0.045 V Atte 1 Harrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning


Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning
O-Level Chemistry | 16 | Qualitative Analysis [1/3]; Author: Bernard Ng;https://www.youtube.com/watch?v=oaU8dReeBgA;License: Standard YouTube License, CC-BY