
EP GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
11th Edition
ISBN: 9780133897340
Author: Petrucci
Publisher: PEARSON CO
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 26, Problem 105SAE
Assign configurations, R or S, to the chiral carbons in the molecules below. Then identity
a. any two identical structures,
b. any two constitutional isomers,
c. any two diastereomers,
d. a pair of enantiomers.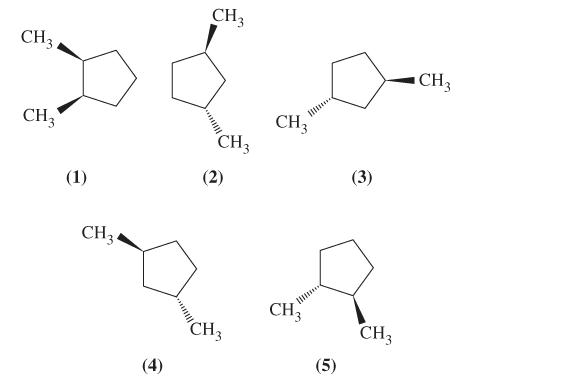
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many chiral centers are in each of the molecules and how to find them?
1. Tell whether the following pairs of structures are identical or not.
2. For those pairs that are not identical, explain what types of isomers they are.
3. If there are chiral centers, denote them with the correct R,S.
H
ÇI
CH3-C-COOH
он
CH3-C-CH2Br
1.
ČI
a
b
81. Consider the structures above. Which of these two contains a
stereocenter? Draw thestructure on your answer sheet and mark the
stereocenter with an asterisk.
82. Which of these two is a chiral molecule? Explain your answer.
83. Which of these two can exist as a pair of enantiomers?
84. Draw the pair of enantiomers using three-dimensional representation.
Chapter 26 Solutions
EP GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
Ch. 26 - Prob. 1ECh. 26 - Draw a structural formula for each of the...Ch. 26 - Prob. 3ECh. 26 - Write structural formulas corresponding to these...Ch. 26 - Prob. 5ECh. 26 - Prob. 6ECh. 26 - Prob. 7ECh. 26 - Prob. 8ECh. 26 - What is the relationship, if any, between the...Ch. 26 - Prob. 10E
Ch. 26 - Prob. 11ECh. 26 - Prob. 12ECh. 26 - Identify the chiral carbon atoms, ¡f any, in the...Ch. 26 - Prob. 14ECh. 26 - Identify the chiral carbon atoms, ¡f any, in the...Ch. 26 - Prob. 16ECh. 26 - Prob. 17ECh. 26 - Prob. 18ECh. 26 - Prob. 19ECh. 26 - By name or formula, give one example of each of...Ch. 26 - Prob. 21ECh. 26 - Prob. 22ECh. 26 - Prob. 23ECh. 26 - Prob. 24ECh. 26 - Prob. 25ECh. 26 - Prob. 26ECh. 26 - Prob. 27ECh. 26 - Prob. 28ECh. 26 - Prob. 29ECh. 26 - Prob. 30ECh. 26 - Prob. 31ECh. 26 - Prob. 32ECh. 26 - Prob. 33ECh. 26 - Prob. 34ECh. 26 - Does each of the following names convey sufficient...Ch. 26 - Prob. 36ECh. 26 - Prob. 37ECh. 26 - Supply condensed structural formulas for the...Ch. 26 - Prob. 39ECh. 26 - Prob. 40ECh. 26 - Classify the carbon atoms in, a. methylbutane, and...Ch. 26 - Classity the carbon atoms in a....Ch. 26 - Prob. 43ECh. 26 - Draw Newman projections for the staggered and...Ch. 26 - Draw the most stable conformation for the molecule...Ch. 26 - Prob. 46ECh. 26 - Prob. 47ECh. 26 - Prob. 48ECh. 26 - Prob. 49ECh. 26 - Prob. 50ECh. 26 - Prob. 51ECh. 26 - Prob. 52ECh. 26 - Prob. 53ECh. 26 - Prob. 54ECh. 26 - Prob. 55ECh. 26 - Prob. 56ECh. 26 - Draw suitable structural formulas to show that...Ch. 26 - Which of the following pairs of molecules are...Ch. 26 - Prob. 59ECh. 26 - Prob. 60ECh. 26 - Name the following molecules with the appropriate...Ch. 26 - Name the following molecules with the appropriate...Ch. 26 - Name the following molecules with the appropriate...Ch. 26 - Prob. 64ECh. 26 - Draw the structure for each of the following. a....Ch. 26 - Prob. 66ECh. 26 - Prob. 67ECh. 26 - Prob. 68ECh. 26 - Prob. 69ECh. 26 - Prob. 70ECh. 26 - Prob. 71ECh. 26 - Prob. 72ECh. 26 - Prob. 73ECh. 26 - Prob. 74ECh. 26 - Supply condensed or structural formulas for the...Ch. 26 - Prob. 76IAECh. 26 - Prob. 77IAECh. 26 - Prob. 78IAECh. 26 - Prob. 79IAECh. 26 - Prob. 80IAECh. 26 - Combustion of a 0.1908 g sample of a compound gave...Ch. 26 - Prob. 82IAECh. 26 - In the monochiorination of hydrocarbons, a...Ch. 26 - A particular colorless organic liquid is known to...Ch. 26 - Prob. 85IAECh. 26 - Give the systematic names, including any...Ch. 26 - Prob. 87IAECh. 26 - Prob. 88IAECh. 26 - Levomethadyl acetate (shown below) is used in the...Ch. 26 - Thiamphenicol (shown below) is an antibacterial...Ch. 26 - Prob. 91IAECh. 26 - Prob. 92IAECh. 26 - Prob. 93IAECh. 26 - Prob. 94IAECh. 26 - Prob. 95IAECh. 26 - For each of the following molecules (a) draw the...Ch. 26 - Prob. 97FPCh. 26 - Prob. 98SAECh. 26 - Explain the important distinctions between each...Ch. 26 - Describe the characteristics of each of the...Ch. 26 - The compound isoheptane is best represented by the...Ch. 26 - Prob. 102SAECh. 26 - Prob. 103SAECh. 26 - Prob. 104SAECh. 26 - Assign configurations, R or S, to the chiral...Ch. 26 - Consider the following pairs of structures In each...Ch. 26 - Prob. 107SAECh. 26 - Prob. 108SAECh. 26 - Prob. 109SAECh. 26 - Prob. 110SAECh. 26 - Prob. 111SAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Estradiol is a female hormone with the following structure: How many chiral carbon atoms are in estradiol?arrow_forwardRefer to the following molecule: CH,CH, Br H. H,C NO2 How many achiral carbons are there? Select ] The molecule is chiral. [ Select] What is the configuration on C2? [Select ] Does the molecule have a meso isomer? [ Select ] How many stereoisomers does the molecule have (including the one already shown)? [ Select ] How many diastereomer pairs does the molecule have? [Select] If the -NO2 and -Br switched places, the new compound will be [Select] with the original compound. What is the third priority substituent on carbon 2? Select]arrow_forwardSketch this molecule and then draw a circle around all the chiral carbons that you can find. 1. Classify the type of isomerism (if any) shown by the compounds below.arrow_forward
- Identify which of the compound is chiral.a. T-butyl alcoholb. 2,2-dimethylpropanec. 1,2,4,5-tetramethylcyclohexaned. 2-hydroxypropanoic acidarrow_forward15. Build two different models of 2-butene. This is a four-carbon chain with a double bond between carbons 2 and 3. a. These two models are stereoisomers. What is their relationship? Enantiomers, diastereomers, something else? b. Classify the two models as cis- and trans-arrow_forwardHow many chiral center does the molecule below have?arrow_forward
- How many chiral canters do we have in the structure?arrow_forward9) There are 3 different cyclopropane molecules with the formula GHĄC12. a. Draw and build the 3 molecules. b. Below each drawing, name each molecule with correct nomenclature. Label a pair that are constitutional isomers. d. Label a pair that are stereoisomers (or configurational isomers). С.arrow_forward1. In the structure provided choose the option YES or NO to indicate if the carbon atoms are chiral. H 2. Assign the absolute configuration of the asymmetric/chiral centers in the provided structures a, b and c. a. a. C-H HO-CH3 H b. NH₂ 3. For the following two structures compared in parts A and B, determine whether they are enantiomers OR diastereomers OR the same structure drawn differently (non-bonding electrons not included for clarity). d. Br Br OH door OH Br b. CH3 HC OH H-C O Br $R= C. OH Br Br OH Br 1arrow_forward
- 1b. Draw ALL isomers (including all stereisomers) of trichlorocyclopentane. Indicate which ones are chiral.arrow_forwardOptical Isomers Optical isomers have a chiral carbon atom. A chiral (or asymmetric) carbon atom is one that has four different groups attached to it. a H This carbon is chiral because it has four different groups attached to it. H,N CH, OH C - Identify which carbon (number) has the chiral center CI OH CH3 H H. H2N H- C-H H C F HICI Harrow_forwardQUESTION #2 Draw all three stereoisomers of the compound shown below. Label each structure you draw according to its relationship to the structure shown here (enantiomer or diastereomer). Label ALL chiral centers on your three molecules as (R) or (S). F. Brarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY