
Concept explainers
(a)
Interpretation:
The E or Z configuration for the following molecule should be determined:
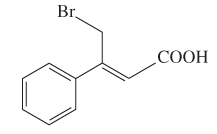
Concept introduction:
(b)
Interpretation:
The E or Z configuration for the following molecule should be determined:
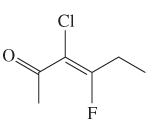
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be named as E and Z-configuration. The E-configuration stands for anti-configuration whereas Z stands for same side configuration. The determination of groups must be done on the basis of their molecular mass. The group or atom with high molecular mass must be numbered as 1 and other with 2. If both 1 numbered group/atom are placed at the same side they will consider as Z-configuration and in E-configuration these groups will be at anti-position.
(c)
Interpretation:
The E or Z configuration for the following molecule should be determined:
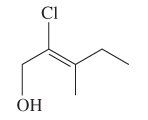
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be named as E and Z-configuration. The E-configuration stands for anti-configuration whereas Z stands for same side configuration. The determination of groups must be done on the basis of their molecular mass. The group or atom with high molecular mass must be numbered as 1 and other with 2. If both 1 numbered group/atom are placed at the same side they will consider as Z-configuration and in E-configuration these groups will be at anti-position.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EP GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
- Cyclopropane and cyclobutane have similar strain energy despite the fact that the C—C—C bond angles of cyclopropane are much smaller than those of cyclobutane. Suggest an explanation for this observation, considering all sources of strain discussed in Chapter 4.arrow_forwardDraw an MO diagram for the following moleculearrow_forwardConsider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.arrow_forward
- Identify the asymmetric carbon in this molecule. Type letter of your choice.arrow_forwardThe correct name for the compound given above is which of the following? Cyclopent-1,3-ene Cyclopent-1,4-ene Cyclopenta-1,3-diene Cyclopenta-1,4-dienearrow_forwardRefer to the equation below to answer questionsWhat is the expected arrangement of the bromine atoms relative to each other among the carbon involvedin pi bonding?A. anti-conformationB. syn-conformationC. trans-configurationD. cis-configurationarrow_forward
- 7. Draw curved arrows indicating the movements of electrons between the following pair of resonance structures. Name the pattern of resonance shown. What is the hybridization of the carbon atoms? 8. Draw the remaining three resonance structures for the molecule in problem 7 above. 9. There are several possible forms of a trisubstituted cyclohexane with the formula C10H200. I have drawn four of them. From these, which one do you think is most commonly naturally occurring, and why? Which is least commonly occurring and why? HO HO" HO HOarrow_forward[Review Topics] [References] Draw two constitutional isomers that share the molecular formula C3H6. Your structures will have the same molecular formula but will have different connectivities.arrow_forward[Review Topics] [References] Calculate Strain energy for the conformer pictured below, using strain energy increments from the table. Strain Energy for Alkanes Interaction / Compound kJ/mol H: H eclipsing H: CH3 eclipsing CH3 CH3 eclipsing gauche butane cyclopropane cyclobutane cyclopentane cycloheptane cyclooctane H₂ H (Calculate your answer to the nearest 0.1 energy unit, and be sure to specify units, kJ/mol or kcal/mol. The answer is case sensitive.) CH3 CH3 H 4.0 5.8 11.0 3.8 115 110 26.0 26.2 40.5 H kcal/mol 1.0 1.4 2.6 0.9 27.5 26.3 6.2 6.3 9.7 Previous Nextarrow_forward
- [Review Topics] [References) Calculate strain energy for the conformer pictured below, using strain energy increments from the table. Strain Energy for Alkanes Interaction / Compound kJ/mol kcal/mol H: H eclipsing 4.0 1.0 H: CH3 eclipsing 5.8 1.4 CH3 : CH3 eclipsing 11.0 2.6 gauche butane 3.8 0.9 cyclopropane 115 27.5 cyclobutane 110 26.3 cyclopentane 26.0 6.2 cycloheptane 26.2 6.3 cyclooctane 40.5 9.7 (Calculate your answer to the nearest 0.1 energy unit, and be sure to specify units, kJ/mol or kcal/mol. The answer is case sensitive.) CH, CH CH3 Next Previous H3C Save and E: 15 tv MacBook Air 80 DII DD F4 F5 F6 F7 F8 F9 F10 F11 F12 $ & ) + 4. 5 7 8. %3D Y [ 11 F J + || < coarrow_forwardFor which isomer would you expect a greater equilibrium percentage of molecules with the alkyl group in the axial position, isopropylcyclohexane or propylcyclohexane? Explain. Isopropylcyclohexane Propylcyclohexanearrow_forwardI need to know the names of these two structurers for my notes but I am not sure how to name them.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
