
(a)
Interpretation:
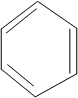
Draw the possible derivatives of benzene ring with molecular formula
Structural or constitutional Isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Benzene is the simplest
(b)
Interpretation:
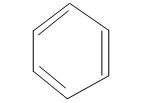
Draw the possible derivatives of benzene ring with molecular formula
Structural or constitutional Isomers can be defined as the molecules with same molecular formula and different structural formula. They are molecules in which the bonded atoms have different bonding with each other although the number of molecules is same. Benzene is the simplest aromatic compound with molecular formula
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EP GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
- Select those compounds that can be correctly called unsaturated and classify each one as an alkene or an alkyne: a.CH3CH2CH3f. b.CH3CH=CHCH3g. c.h.CH2=CHCH2CH3 d.i. e.arrow_forwardConsider a sample of a hydrocarbon at 0.959 atm and 298 K. Upon combusting the entire sample in oxygen, you collect a mixture of gaseous carbon dioxide and water vapor at 1.51 atm and 375 K. This mixture has a density of 1.391 g/L and occupies a volume four times as large as that of the pure hydrocarbon. Determine the molecular formula of the hydrocarbon and name it.arrow_forwardWhat is the difference in bonding and in the general molecular formula between an alkene and an alkane with the same number of carbon atoms?arrow_forward
- Write the molecular formula of each alkane.arrow_forwardWhat is meant by the term “unsaturated hydrocarbon”? What structural feature characterizes unsaturated hydrocarbons?arrow_forwardHow does the structure of a cycloalkane differ from that of a straight-chain or branched-chain alkane?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





