
COLLEGE PHYSICS
2nd Edition
ISBN: 9781464196393
Author: Freedman
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 26, Problem 35QAP
To determine
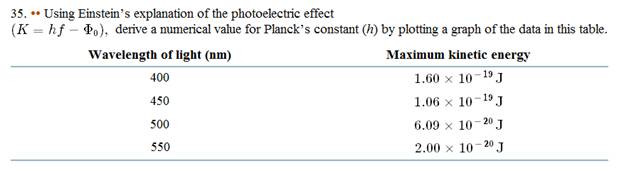
The numerical value of Plank's constant by plotting a graph using the given data.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
•61 SSM The function (x) displayed in Eq. 38-27 can describe a
free particle, for which the potential energy is U(x) = 0 in
Schrödinger's equation (Eq. 38-19). Assume now that U(x) = U, =
a constant in that equation. Show that Eq. 38-27 is a solution of
Schrödinger's equation, with
%3D
-V2m(E – U)
giving the angular wave number k of the particle.
k
k =
Photon Energies and the Electromagnetic Spectrum• Explain the relationship between the energy of a photon in joules or electron volts and its wavelength or frequency.• Calculate the number of photons per second emitted by a monochromatic source of specific wavelength and power.
Sun
0.5 1.0 1.5
Wavelength in micrometers
0.1
2.0
Figure 1-2
Q2 Using Figure 1-2, answer/complete the following:
• In Figure 1-2, what three regions of the EM spectrum are included?
Identify the wavelength at which the Sun emits the most energy:
dmax intensity
Radiation intensity-
Chapter 26 Solutions
COLLEGE PHYSICS
Ch. 26 - Prob. 1QAPCh. 26 - Prob. 2QAPCh. 26 - Prob. 3QAPCh. 26 - Prob. 4QAPCh. 26 - Prob. 5QAPCh. 26 - Prob. 6QAPCh. 26 - Prob. 7QAPCh. 26 - Prob. 8QAPCh. 26 - Prob. 9QAPCh. 26 - Prob. 10QAP
Ch. 26 - Prob. 11QAPCh. 26 - Prob. 12QAPCh. 26 - Prob. 13QAPCh. 26 - Prob. 14QAPCh. 26 - Prob. 15QAPCh. 26 - Prob. 16QAPCh. 26 - Prob. 17QAPCh. 26 - Prob. 18QAPCh. 26 - Prob. 19QAPCh. 26 - Prob. 20QAPCh. 26 - Prob. 21QAPCh. 26 - Prob. 22QAPCh. 26 - Prob. 23QAPCh. 26 - Prob. 24QAPCh. 26 - Prob. 25QAPCh. 26 - Prob. 26QAPCh. 26 - Prob. 27QAPCh. 26 - Prob. 28QAPCh. 26 - Prob. 29QAPCh. 26 - Prob. 30QAPCh. 26 - Prob. 31QAPCh. 26 - Prob. 32QAPCh. 26 - Prob. 33QAPCh. 26 - Prob. 34QAPCh. 26 - Prob. 35QAPCh. 26 - Prob. 36QAPCh. 26 - Prob. 37QAPCh. 26 - Prob. 38QAPCh. 26 - Prob. 39QAPCh. 26 - Prob. 40QAPCh. 26 - Prob. 41QAPCh. 26 - Prob. 42QAPCh. 26 - Prob. 43QAPCh. 26 - Prob. 44QAPCh. 26 - Prob. 45QAPCh. 26 - Prob. 46QAPCh. 26 - Prob. 47QAPCh. 26 - Prob. 48QAPCh. 26 - Prob. 49QAPCh. 26 - Prob. 50QAPCh. 26 - Prob. 51QAPCh. 26 - Prob. 52QAPCh. 26 - Prob. 53QAPCh. 26 - Prob. 54QAPCh. 26 - Prob. 55QAPCh. 26 - Prob. 56QAPCh. 26 - Prob. 57QAPCh. 26 - Prob. 58QAPCh. 26 - Prob. 59QAPCh. 26 - Prob. 60QAPCh. 26 - Prob. 61QAPCh. 26 - Prob. 62QAPCh. 26 - Prob. 63QAPCh. 26 - Prob. 64QAPCh. 26 - Prob. 65QAPCh. 26 - Prob. 66QAPCh. 26 - Prob. 67QAPCh. 26 - Prob. 68QAPCh. 26 - Prob. 69QAPCh. 26 - Prob. 70QAPCh. 26 - Prob. 71QAPCh. 26 - Prob. 72QAPCh. 26 - Prob. 73QAPCh. 26 - Prob. 74QAPCh. 26 - Prob. 75QAPCh. 26 - Prob. 76QAPCh. 26 - Prob. 77QAPCh. 26 - Prob. 78QAPCh. 26 - Prob. 79QAPCh. 26 - Prob. 80QAPCh. 26 - Prob. 81QAPCh. 26 - Prob. 82QAPCh. 26 - Prob. 83QAPCh. 26 - Prob. 84QAPCh. 26 - Prob. 85QAPCh. 26 - Prob. 86QAPCh. 26 - Prob. 87QAPCh. 26 - Prob. 88QAPCh. 26 - Prob. 89QAPCh. 26 - Prob. 90QAPCh. 26 - Prob. 91QAPCh. 26 - Prob. 92QAPCh. 26 - Prob. 93QAPCh. 26 - Prob. 94QAPCh. 26 - Prob. 95QAPCh. 26 - Prob. 96QAPCh. 26 - Prob. 97QAPCh. 26 - Prob. 98QAPCh. 26 - Prob. 99QAPCh. 26 - Prob. 100QAPCh. 26 - Prob. 101QAPCh. 26 - Prob. 102QAPCh. 26 - Prob. 103QAPCh. 26 - Prob. 104QAP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- 3. Dimensional analysis can provide insight into Stefan-Boltzmann's law for the radiation from a black body. According to this law the intensity of radiation, in units of J s- m-2, from a body at temperature T isarrow_forwardQ4) Determine the centripetal force on the Hydrogen atom electron with a wavelength (3.3* 101º m).arrow_forward3. دیا Calculate the wavelength (in nm) of a photon whose energy is 3.5 eV. Given, Planck's constant is 6.63 x 10-34 J-s, Speed of light in a vacuum is 3.0 × 108 m/s and 1 eV 1.602 x 10-1⁹ Jarrow_forward
- The radiant energy from the sun reaches its maximum at a wavelength of about 500.0 nm. What is the approximate temperature of the sun’s surface?arrow_forward(a) How far away must you be from a 650-kHz radio station with power 50.0 kW for there to be only one photon per second per square meter? Assume no reflections or absorption, as if you were in deep outer space. (b) Discuss the implications for detecting intelligent life in other solar systems by detecting their radio broadcasts.arrow_forwardc) Max Planck proposed his constant h to relate the energy of a photon to its frequency. Calculate the energy in eV of red light of wavelength 620 nm.arrow_forward
- 52. • Structure of a virus. To investigate the structure of BIO extremely small objects, such as viruses, the wavelength of the probing wave should be about one-tenth the size of the object for sharp images. But as the wavelength gets shorter, the energy of a photon of light gets greater and could damage or destroy the object being studied. One alternative is to use elec- tron matter waves instead of light. Viruses vary considerably in size, but 50 nm is not unusual. Suppose you want to study such a virus, using a wave of wavelength 5.00 nm. (a) If you use light of this wavelength, what would be the energy (in eV) of a single photon? (b) If you use an electron of this wavelength, what would be its kinetic energy (in eV)? Is it now clear why matter waves (such as in the electron microscope) are often preferable to electromagnetic waves for studying microscopic objects?arrow_forward16. || At what speed is an electron's de Broglie wavelength (a) 1.0 pm, (b) 1.0 nm, (c) 1.0 um, and (d) 1.0 mm?arrow_forward2. When a certain surface is illuminated with light of different wavelengths the following stupping poleritials are observed. Wavelength (nm) Potentlal (V) 366 1.48 405 1.15 436 0.93 492 0.62 546 0.36 579 0.24 Determine by plotting appropriate graphs using the wavelength and potentlal: a) Photoelectric work functlon of the materlal by using the photoelectric equatlon that was given in class. b) Planck's constant given e=1.6x10 Pc.arrow_forward
- •36 If d = 31- 2j + 4k and dz = -5i + 2j-k, then what is %3D ¿lpr x 'p)-(p + 'p)arrow_forward|In a photoelectric effect experiment, the frequency of photons bombarding the surface is increased until photoelectrons just start to leave the surface. If this occurs at a frequency of 6.0 x 1014 Hz, what is the work function of the surface?arrow_forward4) Estimate the surface energies of PE and PET using the Zisman approach. Hint: plot cosine of the contact angle versus surface energy of the probe liquid. YLv Ysv YsL Probe Liquid Y, in mJ/m² OLPE in deg. OL-PET in deg. 1-bromonaphthalene 44.0 dimethyl sulfoxide ethylene glycol diiodomethane 13.6 44.1 47.6 --- 48.0 77.2 49.1 50.8 44.1 31.0 formamide 57.5 75.2 56.9 glycerol 63.1 87.2 66.8 water 72.8 95.3 77.1 PE contact angle data take from Kwok et al. The PET contact angle data was taken from Wu et al.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

University Physics Volume 3
Physics
ISBN:9781938168185
Author:William Moebs, Jeff Sanny
Publisher:OpenStax