
Concept explainers
(a)
Interpretation:
The name of the following compound should be determined:
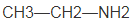
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
(b)
Interpretation:
The name of the following compound should be determined:
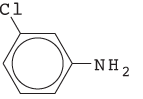
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
(c)
Interpretation:
The name of the following compound should be determined:
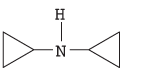
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
(d)
Interpretation:
The name of the following compound should be determined:
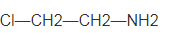
Concept introduction:
The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done such that the structure of organic compound is correctly interpreted from the name.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EBK GENERAL CHEMISTRY
- Write the systematic (IUPAC) names for the amines. The names should have the format alkanamine. H,C-N-CH-CH, systematic (IUPAC) name: NH–CH, H,C-CH-CH-CH, systematic (IUPAC) name: These compounds are secondary amines.arrow_forwardGive the following amine an IUPAC name: CH;-N-CH3 CH3-CH-CH3 Name:arrow_forwardName this amine. CH2-CH2-CH-CH2-CH3 NH₂arrow_forward
- Write the systematic (IUPAC) names for the amines. The names should have the format alkanamine.arrow_forwardAmines are derivatives of oxygen. O True O Falsearrow_forwardIdentify the correct IUPAC name * (CH3CH2)2NCH3 = N-Ethyl, N- methylethanamine (CH3)3CNH2 = 2-methylpropan-2- amine CH3NHCH (CH3)2 = N-Ethylpropan-2- amine (CH3)2CHNH2 = 2, 2-Dimethyl-N- propanamine None of the abovearrow_forward
- what is the difference between an amine and an amide?arrow_forwardDraw complete reactions for: An esterification of propan-1-ol and ethanoic acid A condensation (or dehydration) using propan-1-amine and butanoic acid A controlled oxidation reaction using butanal. Draw and name all isomers with the formula C5H13N. Indicate if the molecules in (d) are first, second, or tertiary amines.arrow_forwardorganic chemistry 2) Which of the following is a secondary amine?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

