
Concept explainers
(a)
Interpretation:
The E or Z configuration for the following molecule should be determined:
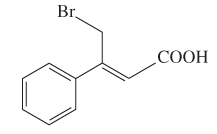
Concept introduction:
(b)
Interpretation:
The E or Z configuration for the following molecule should be determined:
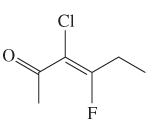
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be named as E and Z-configuration. The E-configuration stands for anti-configuration whereas Z stands for same side configuration. The determination of groups must be done on the basis of their molecular mass. The group or atom with high molecular mass must be numbered as 1 and other with 2. If both 1 numbered group/atom are placed at the same side they will consider as Z-configuration and in E-configuration these groups will be at anti-position.
(c)
Interpretation:
The E or Z configuration for the following molecule should be determined:
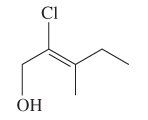
Concept introduction:
Alkenes are unsaturated hydrocarbons with double covalent bond between carbon atoms. On the basis of groups bonded with the double bonded carbon atoms, alkenes can be named as E and Z-configuration. The E-configuration stands for anti-configuration whereas Z stands for same side configuration. The determination of groups must be done on the basis of their molecular mass. The group or atom with high molecular mass must be numbered as 1 and other with 2. If both 1 numbered group/atom are placed at the same side they will consider as Z-configuration and in E-configuration these groups will be at anti-position.
Want to see the full answer?
Check out a sample textbook solution
Chapter 26 Solutions
EBK GENERAL CHEMISTRY
- Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.arrow_forwardFor which isomer would you expect a greater equilibrium percentage of molecules with the alkyl group in the axial position, isopropylcyclohexane or propylcyclohexane? Explain. Isopropylcyclohexane Propylcyclohexanearrow_forwardUse flat representation of rings, not chair in the drawing. Determine the most and least stable. Consider the most stable chair for each of these isomers, and then draw the most stable and least stable isomer based on a comparison of the best chair for each one.arrow_forward
- draw and name two structures that match the description of a trans-dihalocyclopentanearrow_forwardI don't understand how to tell which one goes on which side. When I looked at the answer key to check my answer, I had H/Br and H/NH2 on the wrong sidesarrow_forwardIt is easy to imagine a cyclohexane as a flat hexagon and a lot of the time we draw it that way. Looking at 1,3,5-triethylcyclohexane we cannot tell the stability of the molecule from looking at the flat 2D drawing. Explain why we need to look at the 3D configuration and what conformation (axial,equatorial) would each of the three ethyl groups be in for the most stable configuration.arrow_forward
- Determine which cyclohexane structure has the MOST energy (is the LEAST stable)?arrow_forwardWhich of the following represents the LUMO for cyclopentadiene?arrow_forward.WHICH OF THE COMPOUND SHOW GEOMETRIC ISOMERISM CH3CH=CH2 CH3C(Br)=CH(Br)CH2CH3 CH3CH2CH2CH=CHCH3 HC(CH3)=C(CH3)Cl 2-pentenearrow_forward
- Some of the following examples can show geometric isomerism, and some cannot. Forthe ones that can, draw all the geometric isomers, and assign complete names using theE-Z system. cyclohexene cyclodecenearrow_forwardThese molecules are constitutional isomers.arrow_forwardFor which isomer would you expect a greater equilibrium percentage of molecules with the alkyl group in the axial position, explain.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
