
ORGANIC CHEMISTRY
5th Edition
ISBN: 9781259977596
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 27, Problem 27.25P
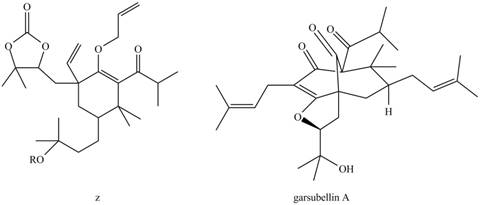
(a) What product is formed by the Claisen rearrangement of compound Z? (b) Using what you have learned about ring-closing metathesis in Chapter 26, draw the product formed when the product in part (a) is treated with Grubbs catalyst. These two reactions are key steps in the synthesis of garsubellin A, a biologically active natural product that stimulates the synthesis of the neurotransmitter acetylcholine. Compounds of this sort may prove to be useful drugs for the treatment of neurodegenerative diseases such as Alzheimer’s disease.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed tosynthesize the drug?
Quinapril (trade name Accupril) is used to treat high blood pressure and congestive heart failure. One step in the synthesis of quinapril involves reaction of the racemic alkyl bromide A with a single enantiomer of the amino ester B. (a) What two products are formed in this reaction? (b) Given the structure of quinapril, which one of these two products is needed to synthesize the drug?
When phenyl 3,4-dimethylbenzoate undergoes electrophilic aromatic substitution, one
ring reacts much more readily than the other. (a) Which one is it? Encircle the ring (b)
Explain your answer. (c) Draw the resulting product/s when it undergoes bromination
reaction.
Product (c)
(a)
Explanation (b)
Chapter 27 Solutions
ORGANIC CHEMISTRY
Ch. 27 - Prob. 27.1PCh. 27 - Problem 27.2
For each molecular orbital in Figure...Ch. 27 - Problem 27.3
(a) Using Figure 27.2 as a guide,...Ch. 27 - Problem 27.4
(a) How many molecular orbitals are...Ch. 27 - Prob. 27.5PCh. 27 - Prob. 27.6PCh. 27 - Prob. 27.7PCh. 27 - Prob. 27.8PCh. 27 - Prob. 27.9PCh. 27 - Prob. 27.10P
Ch. 27 - Problem 27.11
What product would be formed by the...Ch. 27 - Consider cycloheptatrienone and ethylene, and draw...Ch. 27 - Problem 27.13
Show that a thermal suprafacial...Ch. 27 - Prob. 27.14PCh. 27 - a Draw the product of the following [4+2]...Ch. 27 - Prob. 27.16PCh. 27 - Prob. 27.17PCh. 27 - Problem 27.18
Using orbital symmetry, explain why...Ch. 27 - Prob. 27.19PCh. 27 - Prob. 27.20PCh. 27 - Prob. 27.21PCh. 27 - Prob. 27.22PCh. 27 - Prob. 27.23PCh. 27 - Prob. 27.24PCh. 27 - Problem 27.25
(a) What product is formed by the...Ch. 27 - Prob. 27.26PCh. 27 - Prob. 27.27PCh. 27 - Prob. 27.28PCh. 27 - Prob. 27.29PCh. 27 - Prob. 27.30PCh. 27 - Prob. 27.31PCh. 27 - Prob. 27.32PCh. 27 - Prob. 27.33PCh. 27 - Prob. 27.34PCh. 27 - Prob. 27.35PCh. 27 - Prob. 27.36PCh. 27 - Prob. 27.37PCh. 27 - Prob. 27.38PCh. 27 - Prob. 27.39PCh. 27 - Prob. 27.40PCh. 27 - 27.41 What starting materials are needed to...Ch. 27 - Prob. 27.42PCh. 27 - Prob. 27.43PCh. 27 - Prob. 27.44PCh. 27 - Prob. 27.45PCh. 27 - Prob. 27.46PCh. 27 - 27.47 What product is formed from the [5,5]...Ch. 27 - Prob. 27.48PCh. 27 - 27.49 Draw structures for A, B, and C in the...Ch. 27 - Prob. 27.50PCh. 27 - Prob. 27.51PCh. 27 - 27.52 Draw the products of each reaction.
c....Ch. 27 - Prob. 27.53PCh. 27 - 27.54 Draw a stepwise, detailed mechanism for the...Ch. 27 - Prob. 27.55PCh. 27 - Prob. 27.56PCh. 27 - Prob. 27.57PCh. 27 - 27.58 Draw a stepwise, detailed mechanism for the...Ch. 27 - Prob. 27.59PCh. 27 - Prob. 27.60PCh. 27 - Prob. 27.61PCh. 27 - Prob. 27.62PCh. 27 - Prob. 27.63P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Linalool (the Chapter 9 opening molecule) and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can’t linalool be prepared by a similar pathway?arrow_forwardA key step in the synthesis of β-vetivone, a major constituent of vetiver, a perennial grass found in tropical and subtropical regions of the world, involved the reaction of compound A and dihalide B with two equivalents of LDA to form C. Draw a stepwise mechanism for this reaction. β-Vetivone contains a spiro ring system—that is, two rings that share a single carbon atom.arrow_forwardLinalool and lavandulol are two of the major components of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandulol be formed by reduction of a carbonyl compound? (c) Why can't linalool be prepared by a similar pathway?arrow_forward
- We have seen that the alpha carbon atom of an enamine can function as a nucleophile in a Michael reaction, and in fact, enamines can function as nucleophiles in a wide variety of reactions. For example, an enamine will undergo alkylation when treated with an alkyl halide. Draw the structure of intermediate A and the alkylation product B in the following reaction scheme (J. Am. Chem. Soc. 1954, 76, 2029-2030): TSOH -H₂O NH A 1) CHgI 2) H₂O+ B + NH Modify the given copy of the starting material to draw compound A. If needed, use the single bond tool to interconvert between double and single bonds. Edit Drawingarrow_forwardThe compound eutypine is an antibacterial agent isolated from the fungus Eutypa lata. This fungus results in a disease common to vineyards called eutyposis. Give a sequence of reactions that will take the following reactant and give eutypine when the other reactants used in the sequence are acetylene and acetone.arrow_forwardConsider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major product(s) formed when this compound is treated with one equivalent of Br2?arrow_forward
- Consider the tetracyclic compound with rings labeled A–D. (a) Which ring is the most reactive in electrophilic aromatic substitution? (b) Which ring is the least reactive in electrophilic aromatic substitution?arrow_forwardCompound A is first reacted with methylamine in the presence of acid and then treated with NaBH3CN. Using the spectroscopic data given, what is the structure of the product after step 1?arrow_forwardWhich of the follwing is the major product of the reaction sequence shown? (1) NH,NH2, cat. H* (2) КОН, Д NH он NH2 (A) (B) (C) (D) Compound C Compound D Compound B OCompound Aarrow_forward
- What reactant can be used to make this compound by an intramolecular aldol reaction? (A) (B) (C) (D) لسل Harrow_forwardTamoxifen is an estrogen receptor modulator that is used in the treatment of breast cancer. Provide the missing reagents and the structure of compound A in the synthesis of tamoxifen.arrow_forwardQuinapril (trade name Accupril) is used to treat high blood pressure andcongestive heart failure. One step in the synthesis of quinapril involvesreaction of the racemic alkyl bromide A with a single enantiomer of theamino ester B. Given the structure of quinapril, which one of these two products isneeded to synthesize the drug?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License