
(a)
Interpretation:
The structure of the product expected when valine would react with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
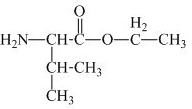
The structure of the product expected when valine would react with
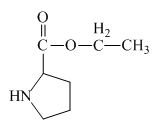
The structure of the product expected when proline would react with
Explanation of Solution
The valine has a carboxylic group in it. This carboxylic group can undergo esterification with ethanol in the presence of sulfuric acid. The corresponding
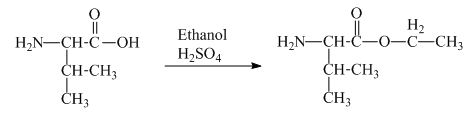
Figure 1
The proline has a carboxylic group in it. This carboxylic group can undergo esterification with ethanol in the presence of sulfuric acid. The corresponding chemical reaction is shown below.
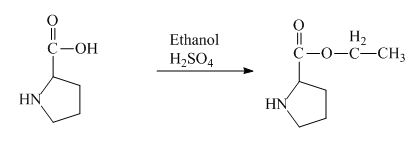
Figure 2
The structure of the product expected when valine would react with
(b)
Interpretation:
The structure of the product expected when valine would react with benzoyl chloride and
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
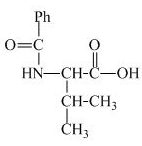
The structure of the product expected when valine would react with benzoyl chloride and ![]() is shown below.
is shown below.
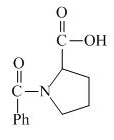
The structure of the product expected when proline would react with benzoyl chloride and
Explanation of Solution
The valine has a basic
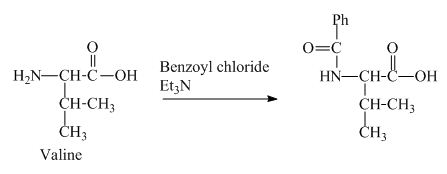
Figure 3
The proline has a basic amine group in it. This group can react with benzoyl chloride to form amide bond. The corresponding chemical reaction is shown below.
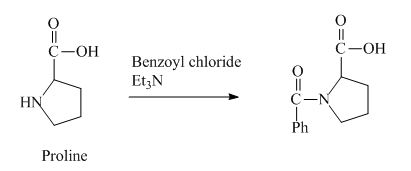
Figure 4
The structure of the product expected when valine would react with benzoyl chloride and
The structure of the product expected when proline would react with benzoyl chloride and
(c)
Interpretation:
The structure of the product expected when valine would react with an aqueous
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with an aqueous
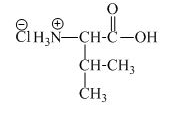
The structure of the product expected when proline would react with an aqueous
Explanation of Solution
The valine has a basic amine group in it. This amine group can react with acid to form an ammonium base. The corresponding chemical reaction is shown below.
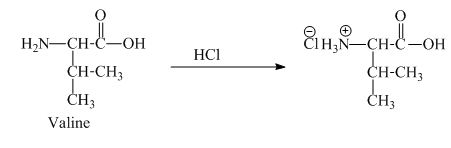
Figure 5
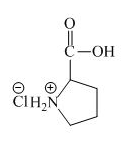
The proline has a basic amine group in it. This amine group can react with acid to form an ammonium base. The corresponding chemical reaction is shown below.
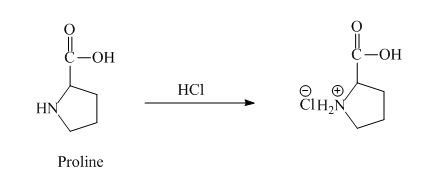
Figure 6
The structure of the product expected when valine would react with an aqueous
The structure of the product expected when proline would react with an aqueous
(d)
Interpretation:
The structure of the product expected when valine would react with an aqueous
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with an aqueous
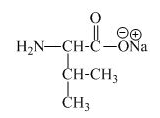
The structure of the product expected when proline would react with an aqueous
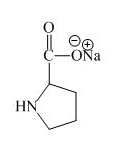
Explanation of Solution
The valine has an acidic carboxylic group in it. This group can react with a base such as sodium hydroxide to form a sodium salt of carboxylic acid. The corresponding chemical reaction is shown below.
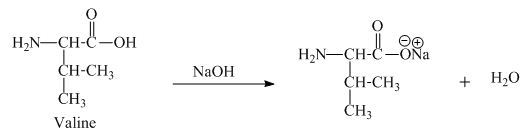
Figure 7
The proline has an acidic carboxylic group in it. This group can react with a base such as sodium hydroxide to form a sodium salt of carboxylic acid. The corresponding chemical reaction is shown below.
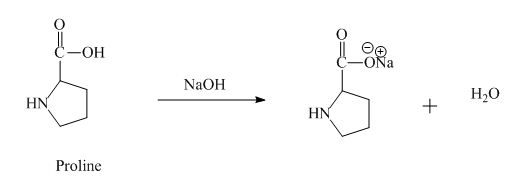
Figure 8
The structure of the product expected when valine would react with an aqueous
The structure of the product expected when proline would react with an aqueous
(e)
Interpretation:
The structure of the product expected when valine would react with aqueous benzaldehyde and
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with aqueous benzaldehyde and
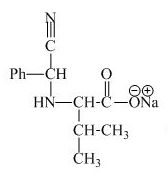
The structure of the product expected when proline would react with aqueous benzaldehyde and
Explanation of Solution
The valine has a basic group in it. This group can be attacked by the benzaldehyde in the presence of sodium cyanide to form the final product. The corresponding chemical reaction is shown below.
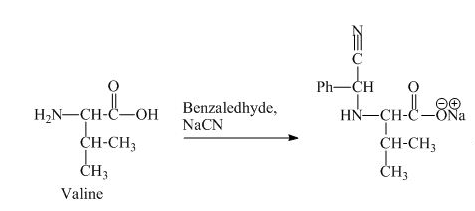
Figure 9
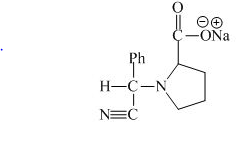
The proline has a basic group in it. This group can be attacked by the benzaldehyde in the presence of sodium cyanide to form the final product. The corresponding chemical reaction is shown below.
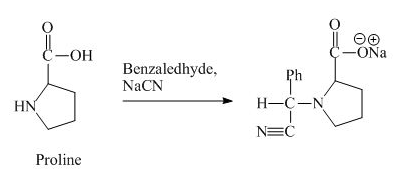
Figure 10
The structure of the product expected when valine would react with aqueous benzaldehyde and
The structure of the product expected when proline would react with aqueous benzaldehyde and
(f)
Interpretation:
The structure of the product expected when valine would react with aqueous Fmoc-![]() followed by neutralizing with
followed by neutralizing with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. The amino acids are classified into acidic, basic, polar, non-polar, essential, and non-essential categories. The three-word abbreviation or one-word abbreviation is used for amino acids.
Answer to Problem 27.43AP
The structure of the product expected when valine would react with aqueous Fmoc-
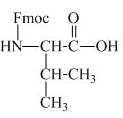
The structure of the product expected when proline would react with aqueous Fmoc-
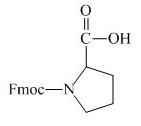
Explanation of Solution
The valine has an amine group. This group can be protected by the reaction of valine with aqueous Fmoc-
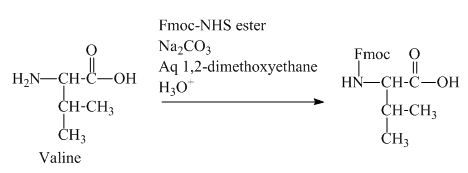
Figure 11
The proline has an amine group. This group can be protected by the reaction of proline with aqueous Fmoc-
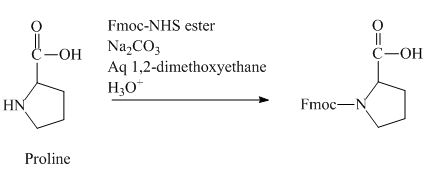
Figure 12
The structure of the product expected when valine would react with aqueous Fmoc-
The structure of the product expected when proline would react with aqueous Fmoc-
(g)
Interpretation:
The structure of the product expected when the given derivative of valine would react with ![]() butyl ester is to be predicted. The structure of the product expected when the given derivative of proline would react with
butyl ester is to be predicted. The structure of the product expected when the given derivative of proline would react with ![]() butyl ester is to be predicted.
butyl ester is to be predicted.
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with ![]() butyl ester is shown below.
butyl ester is shown below.
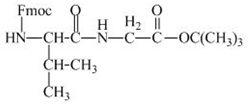
The structure of the product expected when the given derivative of proline would react with ![]() butyl ester is below.
butyl ester is below.
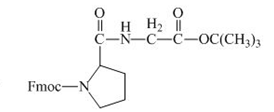
Explanation of Solution
The valine has an amine group. This group can be protected by the reaction of valine with aqueous Fmoc-![]() butyl ester. The corresponding chemical reaction is shown below.
butyl ester. The corresponding chemical reaction is shown below.
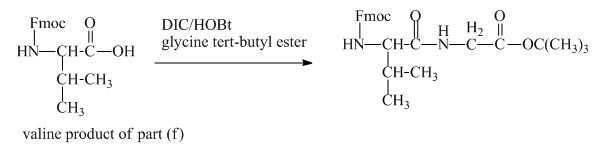
Figure 13
The carboxylic group of the derivative of proline can be protected with the help of reagent with ![]() butyl ester. The corresponding chemical reaction is shown below.
butyl ester. The corresponding chemical reaction is shown below.
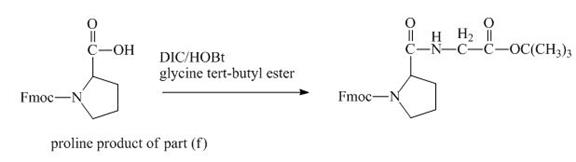
Figure 14
The structure of the product expected when the given derivative of valine would react with ![]() butyl ester is shown in Figure 13.
butyl ester is shown in Figure 13.
The structure of the product expected when the given derivative of proline would react with ![]() butyl ester is shown in Figure 14.
butyl ester is shown in Figure 14.
(h)
Interpretation:
The structure of the product expected when the given derivative of valine would react with anhydrous
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with anhydrous
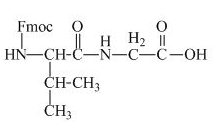
The structure of the product expected when the given derivative of proline would react with anhydrous
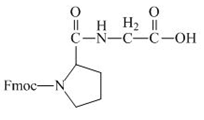
Explanation of Solution
The derivative of valine has an ester linkage in it. The acid,
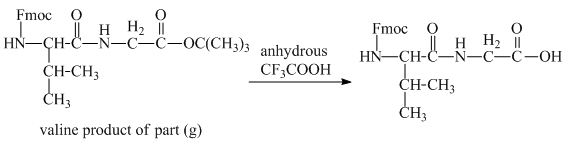
Figure 15
The derivative of proline has an ester linkage in it. The acid,
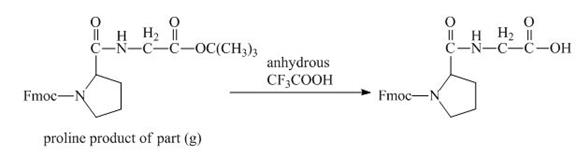
Figure 16
The structure of the product expected when the given derivative of valine would react with anhydrous
The structure of the product expected when the given derivative of proline would react with anhydrous
(i)
Interpretation:
The structure of the product expected when the given derivative of valine would react with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with
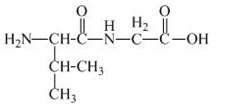
The structure of the product expected when the given derivative of proline would react with
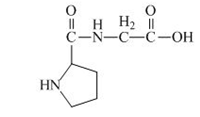
Explanation of Solution
The solution of
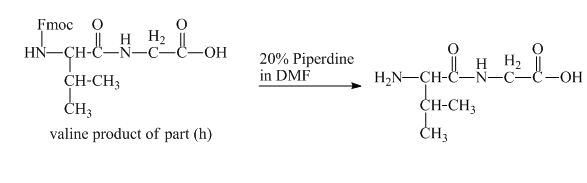
Figure 17
The solution of
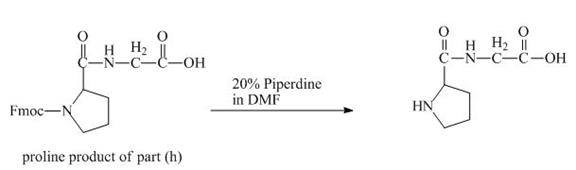
Figure 18
The structure of the product expected when the given derivative of valine would react with
The structure of the product expected when the given derivative of proline would react with
(j)
Interpretation:
The structure of the product expected when the given derivative of valine would react with
Concept introduction:
The chemical compounds in which carbon is bonded with the acidic and basic group along with hydrocarbon side chain are known as amino acids. These functional groups can be protected by different reagent to synthesize peptide chains. The protecting groups can be removed by hydrolysis.
Answer to Problem 27.43AP
The structure of the product expected when the given derivative of valine would react with
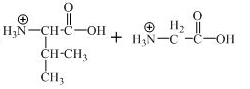
The structure of the product expected when the given derivative of proline would react with
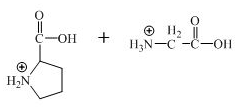
Explanation of Solution
The derivative of valine has amide linkage in it. This amide linkage can be hydrolysis by heating with a strong acid such as
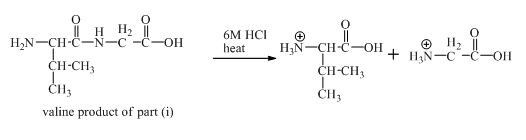
Figure 19
The derivative of valine has amide linkage in it. This amide linkage can be hydrolysis by heating with a strong acid such as
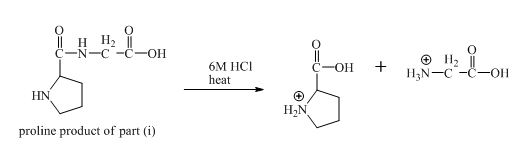
Figure 20
The structure of the product expected when the given derivative of valine would react with
The structure of the product expected when the given derivative of proline would react with
Want to see more full solutions like this?
Chapter 27 Solutions
ORGANIC CHEMISTRY (LL)+ SAPLING ACC >BI
- Propose two structures for a tripeptide that gives Leu, Ala, and Phe on hydrolysis but does not react with phenyl isothiocyanate.arrow_forwardOxytocin, a nonapeptide hormone secreted by the pituitary gland, functions by stimulating uterine contraction and lactation during childbirth. Its sequence was determined from the following evidence: 1. Oxytocin is a cyclic compound containing a disulfide bridge between two cysteine residues. 2. When the disulfide bridge is reduced, oxytocin has the constitution Asn, Cys2, Gln, Gly, Ile, Leu, Pro, Tyr. 3. Partial hydrolysis of reduced oxytocin yields seven fragments: Asp-Cys, Ile-Glu, Cys-Tyr, Leu-Gly, Tyr-Ile-Glu, Glu-Asp-Cys, and Cys-Pro-Leu. 4. Gly is the C-terminal group. 5. Both Glu and Asp are present as their side-chain amides (Gln and Asn) rather than as free side-chain acids. What is the amino acid sequence of reduced oxytocin? What is the structure of oxytocin itself?arrow_forwardIsoleucine and threonine are the only two amino acids with two chirality centers. Assign R or S configuration to the methyl-bearing carbon atom of isoleucine.arrow_forward
- The peptide Proline-Serine-Alanine-Phenylalanine-Glutamine is present at pH 7. Draw the peptide and include stereochemistry.arrow_forwardPrepare valine from the corresponding: a) aldehyde, b) halocarboxylic acid, c) keto acid, d) nitro acid.arrow_forwardExplain why, when the guanidino group of arginine is protonated, the double-bonded nitrogen is thenitrogen that accepts the proton. 2 H+ + NH2NHCH2CH2CH2CHNH +NH3CO−O CO−O H2NC NHCH2CH2CH2CHarrow_forward
- Treatment of a new protein with dansyl chloride reveals two (2) dansyl-labelled derivatives of amino acids, alanine and methionine. What can you deduce about the structure of the proteinfrom these results?arrow_forwardAn unknown decapeptide was isolated and characterized. Complete hydrolysis of this peptide gave : F(2), A,G,C,K,N,T, W and V. Treatment with carboxypeptidase releases A. Reaction with Edman’s reagent gave PTH-T and a nonapeptide. The nonapeptide was treated with trypsin and gave 2 peptides: (V-C-G-A) and (N-FF-W-K). Give the sequence of amino acid in the decapeptide.arrow_forwardAspartame (Nutrasweet®) is a remarkably sweet-tasting dipeptide ester. Complete hydrolysis of aspartame gives phenylalanine, aspartic acid, and methanol. Mild incubation with carboxypeptidase has no effect on aspartame. Treatment ofaspartame with phenyl isothiocyanate, followed by mild hydrolysis, gives the phenylthiohydantoin of aspartic acid.Propose a structure for aspartame.arrow_forward
- Which has a higher percentage of negative charge at physiological pH (7.4), leucine with pI = 5.98 or asparagine with pI = 5.43?arrow_forwardIn the pyrimidine degradative pathway, all pyrimidines undergo conversion to uracil, which undergoes an NADPH-dependent reduction. Show plausible reactions leading from cytidine, cytosine, and uridine to uracil, and write a plausible structure for the product of uracil reduction (dihydrouracil).arrow_forwardValine has pKa's of 2.286 and 9.719. Estimate the fractional composition of Valine in the -1 form at pH=6.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


