
Concept explainers
(a)
Interpretation:
To identify the reactant which reacts with dilute hydrochloric acid
Concept introduction:
Nucleophiles are those species which are electron rich in nature. They are negatively charged. Carboxylate ion is a very good nucleophile. So, it can reacts with dilute
(b)
Interpretation:
To identify the reactant that undergoes hydrolysis.
Concept introduction:
The structure of ester is,
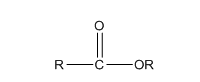
The ester undergoes hydrolysis reaction to form
(c)
Interpretation:
To identify the reactant that neutralizses dilute NaOH.
Concept introduction:
The reaction of an acid and base is called neutralization reaction. The products of neutralization reaction are salt and water. So, the reactant that can neutralize
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
General Chemistry: Principles And Modern Applications Plus Mastering Chemistry With Pearson Etext -- Access Card Package (11th Edition)
- Write the products of the reaction of diphenhydramine (a base) with the acid HCl shown below. Are the reactants or products more soluble in water? Briefly explain.arrow_forwardIdentify the organic functional group of the reactant and the reaction type then predict the functional group(s) of the product(s).The reactant is a(n)a. aromatic ketoneb. aromatic aldehydec. aromatic amided. aromatic alcohole. phenolf. aromatic esterg. aromatic carboxylic acidThe reaction type isa. hydrolysis (in acid)b. hydrolysis (in base)c. amide synthesisd. esterificatione. hydrationf. dehydrationThe product should be a(n)a. carboxlyate ion and aromatic amineb. carboxylate ion and ammonium ionc. carboxylic acid and aromatic alcohold. carboxylic acid and phenole. carboxylic acid and phenoxide ionf. carboxylic ion and aromatic alcoholarrow_forwardwhich has the greatest acidity? a. 2-hexanol b. valeric acid c. naphtol d. ethyl propionate which produces effervescence when dissolved in 10% sodium bicarbonate? a. 2-hexanol b. valeric acid c. napththol d. ethyl propionatearrow_forward
- Calcium carbonate and lactic acid used to prepare Calcium lactate mixture. Whu?arrow_forward7. Strong base is added to the following structure/solution until the reaction is complete. Predict the odor of the NEW product formed /after the reaction has reached completion. Give the name of the reaction. H. H-C-C H. OH OA Sweet smelling Esterification OB. Unpleasant smell; Acid hydrolysis OC Pungent odor; Reduction of carboxylic acids O D. No odor, Neutralizationarrow_forwardAmide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forward
- Identify the reaction type. a. acid base b. dehydration c. hydration d. amide synthesis e. hydrolysis (in base) f. esterification g. hydrolysis (in acid)arrow_forwardDraw the following reactions and label it properly 1. stearic acid reaction with water chemical equation 2. Reaction with NaOH with benzoic acid 3. Reaction with NaOH with acetic acid 4. Reaction with NaOH with stearic acid 5. Reaction with sodium carbonate with acetic acidarrow_forwardthank you 4. Complete the following Reactions b. CH3COOH + NaOHarrow_forward
- Draw the structure of the ff. aldehydes and ketones. a. 3-chlorobutanal 6. 8,8-dibromo -4-cthylcyclo hexanone C. 2,4-dimetnylpontanone • Draw the structure of the ff. compounds. a. oyclobutanecarboxylic acid b. 3,8-dimcthylpontancdioic acid C. 4-aminopcntanoic acid d. 2-mothylcycloheranecarooxylic acid m- chlorobėnzoic acidarrow_forward2. Differentiate acetic acid from hydrochloric acid in terms of. a. Boiling point b. Acidity c. Solubility in ethyl alcoholarrow_forwardWhat is the product of the following reaction? a. b. c. d. مر Br 1)LDA, -780 2)EtBrarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
