
Concept explainers
a)
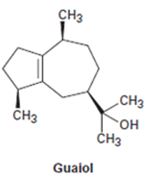
Interpretation:
The chirality centres present in Guaiol are to be indicated by asterisks and the maximum possible number of stereisomers possible is also to be stated.
Concept introduction:
A chirality centre has a carbon attached to four different groups. The maximum number of isomers possible is given by 2n where n is the number of different chiral centres present in the molecule.
To indicate:
The chirality centres present in guaiol are to be indicated by asterisks and to state the maximum possible number of stereisomers for it.
b)
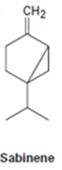
Interpretation:
The chirality centres present in sabinene are to be indicated by asterisks and the maximum possible number of stereisomers possible is also to be stated.
Concept introduction:
A chirality centre has a carbon attached to four different groups. The maximum number of isomers possible is given by 2n where n is the number of different chiral centres present in the molecule.
To indicate:
The chirality centres present in sabinene are to be indicated by asterisks and to state the maximum possible number of stereisomers for it.
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
Organic Chemistry
- The structures of 4 isomers of ketopentose are shown. 1) which of the structures are diastereomers of structure A? C, B, or D 2) which of the structures are enantiomers of structure C? B, A, or D 3) which of the structures are stereoisomers of strcuture A?C, D, or Barrow_forwardDraw all the isomeres of a thioester with molecular formula C4H8OSarrow_forwardIn its pyranose form, naturally occurring D-glucose has five stereocenters. How many possible stereoisomers exist for this structure? a) 5 b) 10 c) 25 d) 32arrow_forward
- Assign R or S configuration to each chirality center in the following molecules.First assign the configuration of the chirality center closest to the top of the Fischer projection. Then give the configuration of the other chirality center.arrow_forwardplease circle or highlight the chiral carbons AND the FOUR DIFFERENT groups of atoms they are bonded to. ( the 11 acetoxy 9 tetrahydrocannabinoic acid Aarrow_forwardd-(-)-Erythrose has the formula HOCH2¬CH(OH)¬CH(OH)¬CHO, and the d in its name implies that it can be degraded to d-(+)-glyceraldehyde. The (-) in its name implies that d-(-)-erythrose is optically active (levorotatory). When d-(-)-erythrose is reduced (using H2 and a nickel catalyst), it gives an optically inactive product of formula HOCH2¬CH(OH)¬CH(OH)¬CH2OH. Knowing the absolute configuration of d-(+)-glyceraldehyde (Section 5-14), determine the absolute configuration of d-(-)-erythrose.arrow_forward
- The cyclic form of this sugar would be A) alpha-anomer as the -CH2OH and the hemiacetal -OH are trans B) beta-anomer as the -CH2OH and the hemiacetal -OH are cis C) beta-anomer as the -CH2OH and the hemiacetal -OH are trans D) alpha-anomer as the -CH2OH and the hemiacetal -OH are cisarrow_forwardDraw and identify (R) and (S) enantiomers of lactic acid, CH3CH(OH)COOH. Use the proper conventions to draw Fischer projections.arrow_forwardDraw the products formed when cholesterol is treated with each reagent. Indicate the stereochemistry around anystereogenic centers in the product.a. CH3COCIb. H2, Pd-Cc. PCCd. leic acid, H+e. [1] BH3 ·THF; [2] H2O2, -OHarrow_forward
- What does chirality mean? What is the difference between diastereomers and enantiomers?arrow_forward2-Butanone is reduced by hydride ion donors, such assodium borohydride (NaBH₄), to the alcohol 2-butanol. Eventhough the alcohol has a chiral center, the product isolated from the redox reaction is not optically active. Explain.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


