
Concept explainers
Interpretation:
It should be explained that why two different products are formed from disrotatory ring closure of (2E, 4Z, 6Z)-octatriene, but only one product is formed from disrotatory ring closure of (2E, 4Z, 6E)-octatriene.
Concept introduction:
Pericyclic reactions are “ any concerted reaction in which bonds are formed or brocken in a cyclic transition state”. There is a single transition state from start to finish, in contrast to a stepwise reaction.
There are mainly three types of pericyclic reactions,
- 1) Electrocyclic reactions
- 2) Cycloaddition reactions
- 3) Sigmatropic reactions
In an electrocyclic reaction “one new sigma- bond is formed or brocken.”
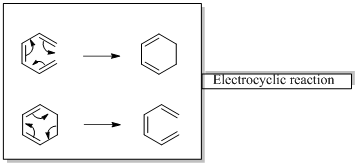
Woodward –Hoffmann rules are the set of rules used to vindicate or predict certain aspects of the stereo chemical outcome and activation energy of pericyclic reactions.
Woodward – Hoffmann rules for Electrocyclic reactions are listed below
Woodward – Hoffmann rules for the configuration of electrocyclic reactions are,
Enantiomers are a type of stereoisomers that have the same molecular formula and constitutions around the atom but differ in their spatial arrangement of groups around the atom.
Trending nowThis is a popular solution!

Chapter 28 Solutions
Organic Chemistry - Access
- Which nucleophile would prefer simple addition over conjugate addition? CH3MgBr NaCN CH3CH2NH2 (CH3CH2)2CuLiarrow_forwardGiven that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related? Recall from Section 3.2A that C6H5 −is a phenyl group, a benzene ring bonded to another group.arrow_forwardGiven that an E2 reaction proceeds with anti periplanar stereochemistry, draw the products of each elimination. The alkyl halides in (a) and (b) are diastereomers of each other. How are the products of these two reactions related? Recall from Section 3.2A that C6H5– is a phenyl group, a benzene ring bonded to another group.arrow_forward
- What is the major E2 elimination product formed from each halide?arrow_forwardWhich set of reagents are most likely to affect the E2 elimination? a. CH3CH2O-, CH3CH2OH b. (CH3)3CO-, (CH3)3COHarrow_forwardExplain why the following bromoketone forms different bicyclic compounds under different reaction conditions:arrow_forward
- Draw the product of the following SN2 reaction, including the stereochemistry at all stereogenic centers. The product of this reaction is aprepitant, a drug used to treat nausea and emesis (vomiting) in chemotherapy patients.arrow_forwardWhich alkyl halide is more reactive in an SN2 reaction with a given nucleophile?arrow_forwardDraw the reaction products for each SN1 reaction and indicate the appropriate stereochemistry.arrow_forward
- Draw a stepwise mechanism for the following reaction that illustrates how two substitution products are formed. Explain why 1-bromohex-2ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reaction conditions, even though it is a 1 ° alkyl halide.arrow_forwardshow an arrow pushing mechanism for the reaction between α-phellandrene and maleic anhydride. Explicitly identify which reactant is the diene and which is the dienophile. Mark the chiral carbon on the α-phellandrene with a *. Draw the 4 possible products from the reaction between α-phellandrene and maleic anhydride. These should include an (R)-endo, (R)-exo, (S)-endo, (S)-exo. Which set of products would be predicted as the major product, endo- or exo-? Explain your reasoning.arrow_forwardDraw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs.a. NaBH4, CH3OHb. [1] LiAlH4; [2] H2Oc. [1] CH3MgBr (excess); [2] H2Od. [1] C6H5Li (excess); [2] H2Oe. Na2Cr2O7, H2SO4, H2Oarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





