
(a)
Interpretation: Bonding and anti-bonding molecular orbitals has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
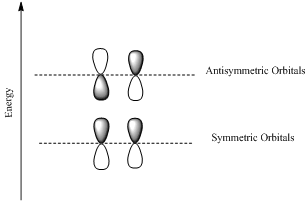
(b)
Interpretation: HOMO and LUMO molecular orbitals in the ground state has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
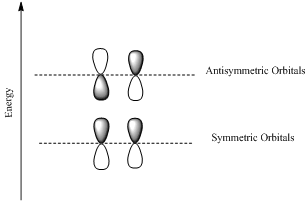
(c)
Interpretation: HOMO and LUMO molecular orbitals in the existed state has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
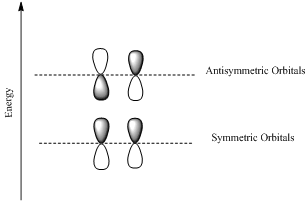
(d)
Interpretation: Symmetric and anti-symmetric MOs has to be determined for the molecular orbitals of
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
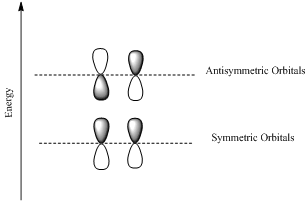
(e)
Interpretation: The relationship between HOMO and LUMO and symmetric and antisymmetric molecular orbitals has to be determined.
Concept introduction:
Molecular orbital theory suggests that atomic orbitals of different atoms combines to create molecular orbitals.
Molecular orbitals can be constructed from linear combination of atomic orbitals.
Bonding orbotals are formed by the additive combination of atomic orbitals and the antibonding orbitals are formed by the substractive combination of atomic orbitals.
Antibonding orbital is a molecular orbital that results when two parallel atomic orbitals with opposite phases interact.
Antibonding orbitals have higher energy than the bonding molecular orbitals.
Ground state and and exited states are the positions with lower and higher energy respectively.
HOMO is a molecular orbital which is the abbrevation of Highest Occupied Molecular Orbital.
LUMO is also a molecular orbital which is the short form of Lowest Unoccupied Molecular Orbital.
If the lobes at the ends of the MO are in phase, then the MO is symmetric.
If the two lobes are out phase then the MO is antisymmetric.
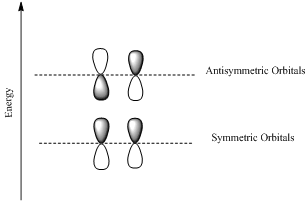
Want to see the full answer?
Check out a sample textbook solution
Chapter 28 Solutions
Organic Chemistry - Access
- The most stable MO of 1,3,5-hexatriene and the most stable MO of benzene Which compound is more stable? Why?arrow_forwardAnswer the following questions regarding the below structures : 1) which form is thermodinamically favored ? 2)For CO stretches in the cis and trans isomers ,do the stretches values give evidence of backbonding? 3)Could these isomers be distinguished by the number of IR active CO stretches (must have dipole change)? How many IR active?arrow_forwardIs the molecular orbital diagram of 1,3-butadiene the same as cyclopenta-1,3-diene? If so how come? Doesn't the latter have 5 p orbitals or will it technically only have 4?arrow_forward
- Answer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?arrow_forwardAnswer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons.Which MOs are bonding? Which are antibonding?arrow_forwardHow would you develop a molecular orbital for a structure like buta-2-diene that has an anion on the first carbon and a cation on the fourth carbon? Also, how would MO look like for 2-dimethyl-1,3-pentadiene?arrow_forward
- Answer the following question for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?arrow_forward1/ Show how the participating p orbitals interact to form the highest energy pi molecular orbital of benzene. 2/ Use the polygon rule to draw the MO energy diagram of the cyclononatetraenyl anion. Assuming planarity, would this ion be aromatic or antiaromatic?arrow_forwardAnswer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons.How many ? -molecular orbitals are there?arrow_forward
- Tips on how to approach a Diels Alder problem like this.arrow_forwardWhat is the best way to draw a molecular orbital diagram for 1,3-cyclopentadiene, identifying the Lowest unoccupied molecular orbital and the highest occupied molecular orbital?arrow_forwardDraw the energy diagram for cyclooctatetraene dianion , C8H8 (2-) , molecular orbitals. The polygon rule is helpful. Label each MO as bonding , antibonding, or nonbonding, and add the nonbonding line. Identify the LOMO, HOMO, LUMO and HUMO orbitals. Is this dianion aromatic , non aromatic or anti aromatic? Thank you!arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

