
Concept explainers
(a)
The ionization energies of the L, M and N shells.
(a)
Answer to Problem 38P
The ionization energies for the L shell is
Explanation of Solution
The figure 1 shows the transitions from higher levels N shell down to K shell.
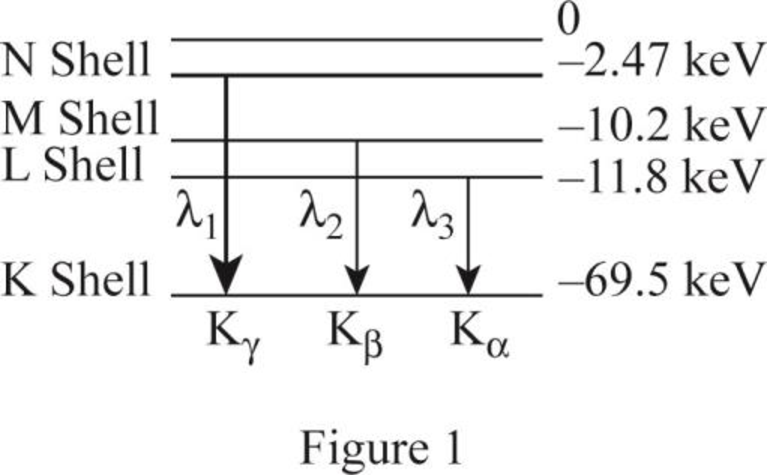
The K series includes the transition from higher levels down to the K shell
The ionization energy for the K shell is
Write the expression for energy of the photon.
Here,
Conclusion:
Substitute
Convert the energy of the photon into kilo electron volt.
Substitute
Convert the energy of the photon into kilo electron volt.
Substitute
Convert the energy of the photon into kilo electron volt.
Similarly the table 1 shows the ionization energy for the shells.
| Photon energy | transition | Energy of level | level | |
| 67.03 | N | |||
| 59.3 | M | |||
| 57.7 | L |
The ionization energy for the K shell is
Therefore, the ionization energies for the L shell is
(b)
Draw the diagram of the transition.
(b)
Answer to Problem 38P
The table 1 shows the transition of the X-ray spectrum of tungsten element.
Explanation of Solution
From (a) The table shows the transition of the X ray spectrum..
| Photon energy | transition | Energy of level | level | |
| 67.03 | N | |||
| 59.3 | M | |||
| 57.7 | L |
Conclusion:
Therefore, the table 1 shows the transition of the X-ray spectrum of tungsten element..
Want to see more full solutions like this?
Chapter 29 Solutions
Bundle: Principles of Physics: A Calculus-Based Text, 5th + WebAssign Printed Access Card for Serway/Jewett's Principles of Physics: A Calculus-Based Text, 5th Edition, Multi-Term
- Derive an expression for the ratio of X-ray photon frequency for two elements with atomic numbers Z1 and Z2.arrow_forwardFor an electron in a hydrogen atom in the n=2 state, compute: (a) the angular momentum; (b) the kinetic energy; (c) the potential energy; and (d) the total energy.arrow_forwardThe yellow light from a sodium-vapor street lamp is produced by a transition of sodium atoms from a 3p state to a 3s state. If the difference in energies of those two states is 2.10 eV, what is the wavelength of the yellow light?arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill
Glencoe Physics: Principles and Problems, Student...PhysicsISBN:9780078807213Author:Paul W. ZitzewitzPublisher:Glencoe/McGraw-Hill University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning





