
Concept explainers
Interpretation:
The pentose phosphate pathway for degrading sugars (see Problem 29-21) is the conversion of ribose 5-phosphate to glyceraldehyde 3-phosphate.
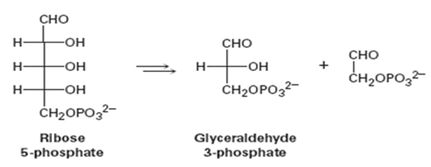
Concept introduction:
Reactions that make and break C-C-Bonds
Reactions that make and break carbon-carbon bonds form the basis of both degradative and biosynthetic
The breakdown of glucose to CO2 involves give such cleavages where as its synthesis involves the reverse process such reactions, considered from the synthetic direction involve addition of a nucleophilic carbanion to an electrophilic carbon atom. The most common electrophilic carbon atoms in such reactions are the sp2 hybridized carbonyl atoms of
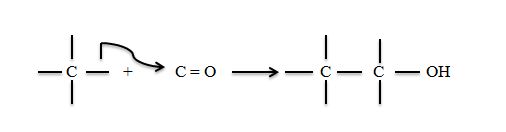
Stabilized carbanions must be generated to add to these electrophilic centres. Three examples are the aldol condensation (catalyzed eg by aldolase) claisen ester condensation (citrate synthase) and the decarboxylation of β-keto acids. (isocitrate dehydrogenase and fatly acid synthase).
Want to see the full answer?
Check out a sample textbook solution
Chapter 29 Solutions
Organic Chemistry
- In the pentose phosphate pathway for degrading sugars, ribulose 5-phosphate is converted to ribose 5-phosphate. Propose a mechanism for the isomerization.arrow_forwardOne of the steps in the pentose phosphate pathway for glucose catabolism is the reaction of xylulose 5-phosphate with ribose 5-phosphate in the presence of a transketolase to give glyceraldehyde 3-phosphate and sedoheptulose 7-phosphate. (a) The first part of the reaction is nucleophilic addition of thiamin diphosphate (TPP) ylide to xylulose 5-phosphate, followed by a retro-aldol cleavage to give glyceraldehyde 3-phosphate and a TPPcontaining enamine. Show the structure of the enamine and the mechanism by which it is formed. (b) The second part of the reaction is addition of the enamine to ribose 5-phosphate followed by loss of TPP ylide to give sedoheptulose 7-phosphate. Show the mechanism.arrow_forwardWhat type of specificity (absolute, group, linkage, or stereochemical) is associated with each of the following enzymes? a. A deaminase b. A phosphatase c. Maltase d. L-Lactate dehydrogenasearrow_forward
- Suggest a name for an enzyme that catalyzes each of the following reactions. a. Hydrolysis of lactose b. Oxidation of nitrite c. Decarboxylation of citrate d. Reduction of oxalatearrow_forwarddraw the chemical structures of the reactant and product of the following reactions: a.) addition of a phosphate group to (ex: phosphorylation of) glycose b.) addition of a phosphate group to (ex: phosphorylation of) fructose-6-phosphate c.) addition of a phosphate group to (ex: phosphorylation of) phosphoenolpyruvatearrow_forwardShow chemical structures for treatment and their mechanism of action for a defective enzyme: Glucose-6-phosphate dehydrogenase.arrow_forward
- Triose phosphate isomerase catalyzes the conversion of dihydroxyacetone phosphate (DHAP) into glyceraldehyde-3-phosphate (G3P). Which of the following illustrates this chemical reaction? a) A b) B c) C d) Darrow_forwardWhat class of enzyme catalyzes each of the following reactions? Hydrolysis of Val-Gly to Val and Gly Transfer of a phosphate from ATP to fructose 6-phosphate Conversion of CH3CH2OH to CH3CHO with NAD+ Book: GENERAL ORGANIC AND BIOLOGICAL CHEMISTRY BY SMITH 4TH EDITIONarrow_forwardThe enzyme triose phosphate isomerase is responsible in which of the following process? A.Formation of fructose 6-phosphate B. Formation of glyceraldehyde 3-phosphate C. Formation of 1,3-bisphosphoglycerate D. Formation of 3-phosphoglycerate E. None of the abovearrow_forward

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




