
Concept explainers
a)
Interpretation:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above, is to be shown.
Concept introduction:
Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To show:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above.
b)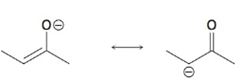
Interpretation:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above, is to be shown.
Concept introduction:
Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To show:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above.
c)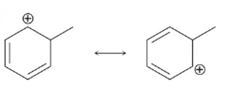
Interpretation:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above, is to be shown.
Concept introduction:
Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To show:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above.
Trending nowThis is a popular solution!

Chapter 2 Solutions
ORGANIC CHEM.(LL)-W/OWL V2 >CUSTOM<
- Define the consequence of resonance stabilization ?arrow_forwardDraw the conjugate base of C5F5H. Include lone pairs of electrons on all atoms. Add non-zero formal charges where appropriate.arrow_forwardUsing the curved arrow formalism, draw two major resonance contributors to the structure of the following molecule. Circle that structure that contributes most to the overall structure.arrow_forward
- All resonance structures with arrow. Major resonance?arrow_forward1. Add nonbonding electron pairs to each atom where it is implied by the line-angle drawings shown below. 2. Use the curved arrow formalism to generate four additional, valid resonance structures for each compound. 3. Rank your resonance structures in order of increasing contribution to the overall nature of the molecule.arrow_forwardCircle all the isolated (not conjugated) pi bond(s)arrow_forward
- Add curved arrows to show how the first resonance structure can be converted into the second.arrow_forwardProvide a mechanism. Illustrate role played by N,N,N-triethylamine in reaction. Use curved arrows to show electron movement and show all non-zero formal charges.arrow_forwardI’m supposed to make a resonance structure of this with the same formal charge and I’ve tried at least 7 times with different combinations but I can’t seem to get it right. Can you show me the general direction of where I should go with this? Thanks!arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
