
Biochemistry
6th Edition
ISBN: 9781305577206
Author: Reginald H. Garrett, Charles M. Grisham
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 18P
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
The acyl-CoA synthetase reaction activates fatly acids for oxidation in cells:
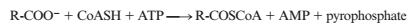
The reaction is driven forward in part by hydrolysis of ATP to AMP and pyrophosphate. However, pyrophosphate undergoes furl her cleavage to yield two phosphate anions. Discuss the energetics of this reaction both in the presence and absence of pyrophosphate cleavage.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Biochemistry
Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at (he end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Prob. 9PCh. 3 - Answers to all problems are at the end of this...
Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Graphical Analysis of MWC Allosteric Enzyme Kinetics (Integrates with Chapter 1.1) Draw both Line weaver-Burk plots and Hanes-Woolf plots for an MWC allosteric enzyme system, showing separate curves for the kinetic response in (a) the absence of any effectors, (b) the presence of allosteric activator Λ, and (c) the presence of allosteric inhibitor I.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Describe the secondary structure of each subdomain of malonyl-CoA: ACP transferase Explain the difference between parallel and antiparallel beta sheets.arrow_forwardAnswers to all problems are at the end οΓthis book. Detailed solutions are available in the Student Solutions Manual. Study Guide, and Problems Book. Using Site-Direcled Muta.nts to Understand an Enzyme Mechanism In this chapter, the exponent in which Craik and Rutter replaced Asp102 with Asn in trypsin (reducing activity 10,000 -fold) was discussed. On the basis of your knowledge of the catalytic triad structure in trypsin, suggest a structure for the “uncatalytic triad of Asn-His-Ser in this mutant enzyme. Explain why the structure you have proposed explains the reduced activity of the mutant trypsin. See the original journal articles (Sprang, et al., 1987. Science 237:905-913) to Craik, et al., 1987. Scieence 237:909-913) to see Craik and Rutter's answer to this question.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Determining the Branch Points and Reducing Ends of Amylopectin A 0.2-g sample of amylopectin was analyzed to determine the fraction of the total glucose residues, that are branch points in the structure. The sample was exhaustively methylated and then digested, yielding 50-mol of 2,3-dimethylgluetose and 0.4 mol of 1,2,3,6- letramethylglucose. What fraction of the total residues are branch points? I low many reducing ends does this sample of amylopectin have?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Graphing the Results from Kinetics Experiments with Enzyme Inhibitors The following kinetic data were obtained for an enzyme in the absence of any inhibitor (1), and in the presence of two different inhibitors (2) and (3) at 5 mM concentration. Assume [ET] is the same in each experiment. Graph these data as Lineweaver-Burk plots and use your graph to find answers to a. and b. a. Determine Vmax and Km for the enzyme. b. Determine the type of inhibition and the K1 for each inhibitor.arrow_forwardAnswers to all problems are at the end οΓthis book. Detailed solutions are available in the Student Solutions Manual. Study Guide, and Problems Book. Comparison of Emzymatic and Nonenzymatic Rate Constants The for alkaline phosphatase—catalyzed hydrolysis of melhylphoiphate is approximately 14/sec at pH 8 and 25ºC. The rate constant for the uncatalyzed hydrolysis of methyl phosphate under the same conditions is approximately I0-15/sec. What is the difference in the free energies of activation of these two reactions?arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Graphical Analysis of Negative Gooperativity in KNF Allosteric Enzyme Kinetics The KNF model for allosteric transitions includes the possibility of negative cooperativity Draw Lineweaver-Burk and Hanes-Woolf plots for the case of negative cooperatively m substrate binding. (As a point of reference, include a line showing the classic Michaelis-Menten response of v to [S].)arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Quantitative Relationships Between Rale Constants to Calculate Km, Kinetic Efficiency (kcat/Km) and Vmax - II Triose phosphate isomerase catalyzes the conversion of glyceraldehyde-3-phosphate to dihydroxy-acetone phosphate. Glyceraldehyde3PdihydroxyacetonePThe Km of this enzyme tor its substrate glyceraldehyde-3-phosphate is 1.8 10-5 M. When [glyceraldehydes-3-phosphate] = 30 M, the rate of the reaction, v, was 82.5 mol mL-1 sec-1. a. What is Vmax for this enzyme? b. Assuming 3 nanomoles per mL of enzyme was used in this experiment ([Etotal]) = 3 nanomol/mL), what is kcat for this enzyme? c. What is the catalytic efficiency (kcat/Km) for triose phosphate isomerase? d. Does the value of kcat/Km reveal whether triose phosphate isomerase approaches catalytic perfection? e. What determines the ultimate speed limit of an enzyme-catalyzed reaction? That is, what is it that imposes the physical limit on kinetic perfection?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Quantitative Relationships Between Rate Constants to Calculate Km, Kinetic Efficiency (kcat/Km) and Vmax - III The citric acid cycle enzyme fumarase catalyzes the conversion of fumarate to form malate. Fumarate+H2OmalateThe turnover number, kcat, for fumarase is 800/sec. The Km of fumarase for its substrate fumarate is 5M. a. In an experiment using 2 nanomole/mL of fumarase, what is Vmax? b. The cellular concentration of fumarate is 47.5 M. What is v when [fumarate] = 47.5 M? c. What is the catalytic efficiency of fumarase? d. Does fumarase approach catalytic perfection?arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Assessing the Formation and Composition of Limit Dextrins Prolonged exposure of amylopectin to starch phosphorylase yields a substance called a limit dextrin. Describe the chemical composition of limit dextrins. and draw a mechanism for the enzyme-catalyzed rcactioa that can begin the breakdown of a limit dextrin.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual. Study Guide, and Problems Book. Assessing the-Metabolic Consequences of Life Without Enzymes The incredible catalytic power of enzymes can perhaps best be appreciated by imagining how challenging life would be without just one of the thousands of enzymes in the human body. For example, consider life without fnnctose-1,6-btsphosphatase, an enzyme in the gluconeogenesis pathway in Liver and kidneys (see Chapter 22). which helps product new glucose from the food we eat: Fructose-1.6-blsphosphate + H2O Fmrlose-6-P + Pi The human brain requires glucose as its only energy source, and the typical brain consumes about 120 g (or 480 kilocalories) of glucose dally. Ordinarily, two pieces of sausage pizza could provide more than enough potential glucose to feed the brain for a day. According to a national fast-food chain, two pieces of sausage pizza provide 1340 kilocalories. 48% of which is from fat. Fats cannot be converted to glucose in gluconeogenesis, so that leaves 697 kilocalories potentially available for glucose synthesis. The first-order rate constant for the hydrolysis of fructose-l.6-bispliosphate in the absence of enzyme is 2 10-20 /sec. Calculate how long it would take to provide enough glucose for one day of brain activity from two pieces of sausage pizza without the enzyme. The following graphs show the temperature and pH dependencies of four enzymes, A, Β, X, and Y. Problems 12 through IS refer to these graphs.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Describe the Effects on cAMP and Glycogen Levels in Cells Exposed to Cholera Toxin Cholera toxin is an enzyme that covalently modifies the G-subunit of G proteins. (Cholera toxin catalyzes the transfer of ADP-ribose from NAD+ to an arginine residue in Gan ADP-ribosylation reaction.) Covalent modification of G� inactivates its GTPase activity. Predict the consequences of cholera to.vin on cellular cAMP and glycogen levels.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning
Chapter 7 - Human Movement Science; Author: Dr. Jeff Williams;https://www.youtube.com/watch?v=LlqElkn4PA4;License: Standard youtube license