
Biochemistry
6th Edition
ISBN: 9781305577206
Author: Reginald H. Garrett, Charles M. Grisham
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 5P
Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book.
Calculating the Effect of pH on
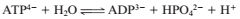
If the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Biochemistry
Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at (he end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Prob. 9PCh. 3 - Answers to all problems are at the end of this...
Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...Ch. 3 - Answers to all problems are at the end of this...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Draw the Titration Curve for a Weak Acid and Determine its pKa from the Titration Curve When a 0.1 M solution of a weak acid was titrated with base, the following results were obtained: Plot the results of this titration and determine the pK a of the weak acid from your graph.arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. CalculatingGandSfromH The equilibrium constant for some process AB 0.5 at 20°C and 10 at 30°C. Assuming that G is independent of temperature, calculate H for this reaction. GandSat20Candat30C Why- is it important in this problem to assume that H is independent of temperature?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Graphical Analysis of MWC Allosteric Enzyme Kinetics (Integrates with Chapter 1.1) Draw both Line weaver-Burk plots and Hanes-Woolf plots for an MWC allosteric enzyme system, showing separate curves for the kinetic response in (a) the absence of any effectors, (b) the presence of allosteric activator Λ, and (c) the presence of allosteric inhibitor I.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Plot the Titration Curve for Bicine and Calculate How to Prepare a pH 7.5 Bicine Buffer Solution Bicine (N, N—bis (2-hydroxyethyl) glycine) is another commonly used buffer in biochemistry labs. The structure of Bicine in its fully protonated form is shown here: Draw the titration curve for Bicine. assuming the pA'a for its free COOH group is 2.3 and the pAa for its tertiary amino group is 8.3. Draw the structure of the fully deprotonated form (completely dissociated form) of bicine. You have available a U.l Msolution of Bicine at its isoelectric point (pH|)T 0.1 M solutions of HCI and NaOH. and ample distilled water. Describe the preparation of 1 L of 0.U4 M Bicine buffer. pH 7.5. What is the concentration of the fully protonated form of Bicine in your final buffer solution?arrow_forwardAnswers to all problems are at the end οΓthis book. Detailed solutions are available in the Student Solutions Manual. Study Guide, and Problems Book. Calculation of Rate Enhancement from Energies of Activation The relationships between the free energy terms defined in the solution to Problem 4 earlier are shown in the following figure. If the energy of the ES complex is 10 kJ/mol lower than the energy of E + S, the value of Ge:is 20 kJ/mol, and the value of Ge:is 90 kJ/mol what is the rate enhancement achieved by an enzyme in this case?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Calculating and Keq for Coupled Reactions For the process A B. Keq (AB) is 0.02 at 370C. For the process B C. Keq (BC)=1000 at 370C. Determine Keq (AC), the equilibrium constant for the overall process A C, from Keq((AB) and (BC). Determine standard-state free energy changes for all three processes, and use G. (AC) to determine Keq (AC). Make sure that ibis value agrees with that determined m part a of this problem.arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Quantitative Relationships Between Rate Constants to Calculate Km, Kinetic Efficiency (kcat/Km) and Vmax - VI The enzyme catalase catalyzes the decomposition of hydrogen peroxide: 2H2O22H2O+O2The turnover number (kcat) for catalase is 40,000,000 sec-1. The Km of catalase for its substrate H2O2 is 0.11 M. a. In an experiment using 3 nanomole/L of catalase, what is Vmax? b. What is v when [H2O2] = 0.75 M? c. What is the catalytic efficiency of catalase? d. Does catalase approach catalytic perfection?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Interpreting Kinetics Experiments from Graphical Patterns The following graphical patterns obtained from kinetic experiments have several possible interpretations depending on the nature of the experiment and the variables being plotted. Give at least two possibilities for each.arrow_forwardAnswers to all problems are at the end οf this book. Detailed solutions are available in the Student Solutions Manual. Study Guide, and Problems Book. Understanding the Very Tight Binding of Transition States Another consequence of tight binding (problem 9) is the free energy change for the binding process. Calculate Gfor an equilibrium with a KD of 10-27 M. Compare this value to the free energies of the noncovalent and covalent bonds with which you are familiar. What are the implications of this number, in terms of the binding of a transition state to an enzyme active site?arrow_forward
- Answers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Calculating [H+] from pH Calculate the following from the pH values given in Table 2.3. [H+] in vinegar [H+] in saliva [H+] in household ammonia [OH-] in milk of magnesia [OH-] in beer [H+] inside a liver cellarrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Determining the Branch Points and Reducing Ends of Amylopectin A 0.2-g sample of amylopectin was analyzed to determine the fraction of the total glucose residues, that are branch points in the structure. The sample was exhaustively methylated and then digested, yielding 50-mol of 2,3-dimethylgluetose and 0.4 mol of 1,2,3,6- letramethylglucose. What fraction of the total residues are branch points? I low many reducing ends does this sample of amylopectin have?arrow_forwardAnswers to all problems are at the end of this book. Detailed solutions are available in the Student Solutions Manual, Study Guide, and Problems Book. Using Graphical Methods to Derive the Kinetic Constants for an Ordered, Single-Displacement Reaction The general rate equation for an ordered, single-displacement reaction where A is the leading substrate is v=Vmax[ A ][ B ](KsAKmB+KmA[ B ]+KmB[ A ]+[ A ][ B ])Write the Lineweaver-Burk (double-reciprocal) equivalent of this equation and from it calculate algebraic expressions for the following: a. The slope b. The y-intercepts c. The horizontal and vertical coordinates of the point of intersection when 1/v is plotted versus 1/[B] at various fixed concentrations of Aarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning
Chapter 7 - Human Movement Science; Author: Dr. Jeff Williams;https://www.youtube.com/watch?v=LlqElkn4PA4;License: Standard youtube license