
Concept explainers
Answer each question about oxycodone, a narcotic analgesic used for severe pain.
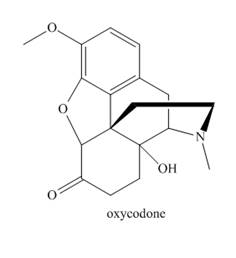
a. Identify the functional groups in oxycodone.
b. Classify any alcohol, amide or
c. Which proton is most acidic?
d. Which site is most basic?
e. What is the hybridization of the N atom?
f. How many
(a)
Interpretation: The functional groups present in the given compound, oxycodone are to be identified.
Concept introduction: An atom or a group of atoms that shows characteristic physical and chemical properties are collectively known as functional groups. The functional group is the most reactive part present in the molecule. The main functional groups are
Answer to Problem 3.61P
The functional groups present in the given compound, oxycodone are shown below.
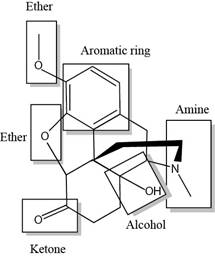
Explanation of Solution
The functional groups present in the given compound, oxycodone are shown as,
Figure 1
Thus, there are total five functional groups present in oxycodone which are ether group, amine group, alcoholic group, ketonic group and aromatic ring as highlighted above.
There are total five functional groups present in oxycodone which are ether group, amine group, alcoholic group, ketonic group and aromatic ring.
(b)
Interpretation: The alcohol, amine or amide groups present in the given compound, oxycodone are to be classified as
Concept introduction: The amine, amide or alcoholic groups are identified as primary
Answer to Problem 3.61P
The classification of alcohol, amine or amide groups present in the given compound, oxycodone as
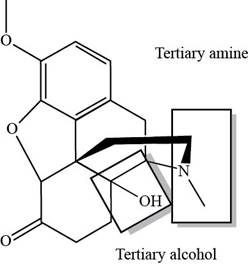
Explanation of Solution
The classification alcohol, amine or amide groups present in the given compound, oxycodone are shown as,
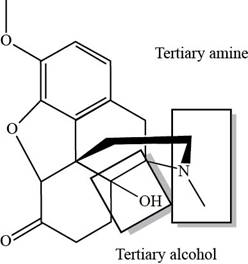
Figure 2
The given compound consists of one tertiary amine and one tertiary alcohol.
The given compound, oxycodone consists of one tertiary amine and one tertiary alcohol.
(c)
Interpretation: The most acidic proton present in the given compound, oxycodone is to be labeled.
Concept introduction: The most acidic hydrogen
Answer to Problem 3.61P
The labeling of the most acidic proton present in the given compound, oxycodone is shown below.
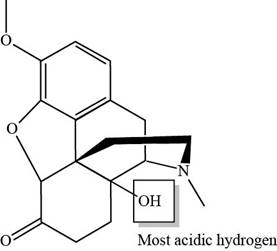
Explanation of Solution
The labeling of the most acidic proton present in the given compound, oxycodone is shown as,

Figure 3
The electronegativity increases while moving left to right in the period. Thus, oxygen is more electronegative than nitrogen atom as it is located at the extreme right position. According to the structure of oxycodone, hydrogen atom that is bonded with oxygen atom is the most acidic one because oxygen is more electronegative than nitrogen atom. The higher electronegativity corresponds to the greater acidic character of hydrogen atom attached to the electronegative atom.
The labeling of the most acidic proton present in the given compound, oxycodone is shown in Figure 3.
(d)
Interpretation: The most basic site in the compound oxycodone is to be labeled.
Concept introduction: According to Bronsted-Lowry theory, the species that easily tends to accept the proton is known as base and the species that easily donate the proton is known as acid. The high delocalization of electron pair corresponds to the high basic character of the atom.
Answer to Problem 3.61P
The labeling of the most basic site in the compound oxycodone is shown below.
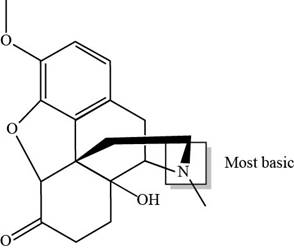
Explanation of Solution
The labeling of the most basic site in the compound oxycodone is shown as,

Figure 4
According to the structure of oxycodone, the site at which amine group is present is the most basic site because in amines, the pair of electrons over nitrogen atom gets highly delocalized. Hence, the most basic site is present at tertiary amine.
The labeling of the most basic site in the compound oxycodone is shown in Figure 4.
(e)
Interpretation: The hybridization of
Concept introduction: The procedure of intermixing of the atomic orbitals in an atom is known hybridization. This intermixing of the atomic orbitals is used to form a set of new atomic orbital with different geometry.
Answer to Problem 3.61P
The hybridization of
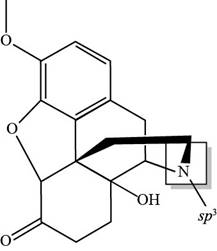
Explanation of Solution
The hybridization of

Figure 5
As it is clearly seen that nitrogen atom present in oxycodone possesses three bond pairs and one lone pair. This type of bonding indicates that N-atom in oxycodone is
The hybridization of
(f)
Interpretation: The total number of
Concept introduction: The procedure of intermixing of the atomic orbitals in an atom is known hybridization. This intermixing of the atomic orbitals is used to form a set of new atomic orbital with different geometry.
Answer to Problem 3.61P
There are total seven
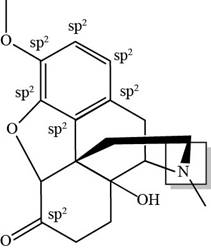
Explanation of Solution
The total number of

Figure 6
There are total seven
The total number of
Want to see more full solutions like this?
Chapter 3 Solutions
Loose Leaf for Organic Chemistry
- For each molecule below, draw the conjugate acid or conjugate base or both if the molecule hasboth a conjugate acid and a conjugate base (e.g., water).arrow_forwardWhy is H2SO4 a weaker acid than HClO4? I. S is less electronegative than Cl. II. H2SO4 has fewer terminal O atoms than HClO4. III. The S–H bond is less polar than the Cl–H bond. Select one: a.I only b.I and II only c.I, II, and III d.II onlyarrow_forward6. Circle the most acidic and put an X over the least acidic in each set of three. (Please help with subparts a-c)arrow_forward
- Dd.59. I need part A and C please......arrow_forward2 part question please help! a) Which of the following is the strongest acid? a. CH3COCH3 (pKa = 19.3) b. CH3COOH (pKa = 4.76) c. CH3(CH2)2CH3 (pKa = 50) d. CH3CH2OH (pKa = 16.00) e. CH3(CH2)3C≡CH (pKa = 25) b) A carbon-carbon double bond contains the following bonding orbitals: a. one π and one π* b. two π c. one σ and one π d. one σ and one π* e. two σarrow_forwardpart A 3arrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



