
Concept explainers
Skill Building Wavelength, Frequency, and Energy
Return to Figure 3.4, and calculate the amount of energy present in a wave of red light and a wave of blue light. Are they the same amount of energy? Why or why not?
In the not-too-distant past, many people used film exposure to take pictures. A “dark” room was a place where this film was developed into the actual images based on how light exposed the film during the taking of the pictures. These dark rooms used red lights during the time of developing the film because a special dye was added to the film that was not affected by light with a wavelength between 620 and 750 nm. Do you think these rooms could have also used blue lights? Why or why not?
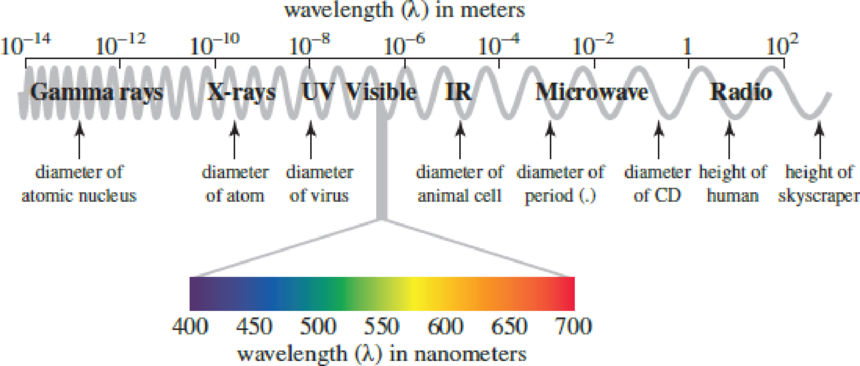
Figure 3.4
The
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Chemistry In Context
Additional Science Textbook Solutions
Organic Chemistry
Essential Organic Chemistry (3rd Edition)
Introductory Chemistry (6th Edition)
Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry: Structure and Properties
- The frequency of the strong red line in the spectrum of potassium is 3.91 1014/s. What is the wavelength of this light in nanometers?arrow_forwardAn atom has a line spectrum consisting of a red line and a blue line. Assume that each line corresponds to a transition between two adjacent energy levels. Sketch an energy-level diagram with three energy levels that might explain this line spectrum, indicating the transitions on this diagram. Consider the transition from the highest energy level on this diagram to the lowest energy level. How would you describe the color or region of the spectrum corresponding to this transition?arrow_forwardThe element cesium was discovered in 1860 by Robert Bunsen and Gustav Kirchhoff, who found two bright blue lines in the spectrum of a substance isolated from a mineral water. One of the spectral lines of cesium has a wavelength of 456 nm. What is its frequency?arrow_forward
- Laser light of a specific frequency falls on a crystal that converts a portion of this light into light with double the original frequency. How is the wavelength of this frequency-doubled light related to the wavelength of the original laser light? Suppose the original laser light was red. In which region of the spectrum would the frequency-doubled light be? (If this is in the visible region, what color is the light?)arrow_forwardLook at the following orbital diagrams and electron configurations. Which are possible and which are not, according to the Pauli exclusion principle? Explain. a b c d 1s22s22p4 e 1s22s42p2 f 1s22s22p63s23p103d10arrow_forwardRepresent the transfer of electrons from magnesium to oxygen atoms to assume noble-gas configurations. Use Lewis electron-dot symbols.arrow_forward
- Exercise 4.4 Give the symbol for the phosphorus atom (Z = 15) that contains 17 neutrons.arrow_forwardGive the orbital diagram and electron configuration for the carbon monoxide molecule, CO. What is the bond order of CO? Is the molecule diamagnetic or paramagnetic?arrow_forwardEXERCISE 3 Calculate the frequency at which the transition J= 5 ¬ J=4 will appear in the rotation spectrum of 14Ν16Ο. The bond length of the molecule is 115 pm. Use the following atomic mass data: a) N: 14 amu, b) O: 16 amuarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

