
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 3.8, Problem 28P
Rank the following compounds in each set from most soluble to least soluble in water:
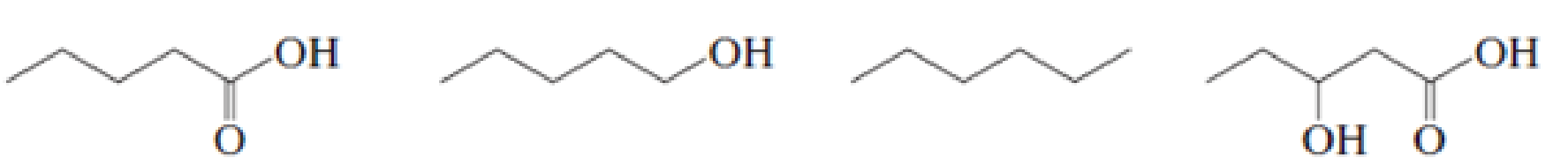

Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. which compound is more soluble in water: CH3 (CH2)3 COO- Na+ Or
CH3(CH2)3 COOH
2. which compound is more soluble in an organic solvent:
CH3 (CH2)3 COO- Na+ Or
CH3(CH2)3 COOH
Rank the following compounds in order of increasing water solubility.
4-Methylphenol, CH3C6H4OH (pKa 10.26), is only slightly soluble in water, but its sodium salt, CH3C6H4O- Na+, is quite soluble in water. In which solution(s) will 4-methylphenol dissolve?
Q.) Aqueous Na2CO3
Chapter 3 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 3.1 - Name each of the following:Ch. 3.1 - Draw the structures and name the four...Ch. 3.1 - Draw the structure for each of the following...Ch. 3.1 - Name the following compounds:Ch. 3.1 - Prob. 7PCh. 3.2 - What is each compounds systematic name?Ch. 3.2 - Prob. 11PCh. 3.3 - Prob. 12PCh. 3.3 - Convert the following condensed structures into...Ch. 3.3 - The molecular formula for ethyl alcohol (CH3CH2OH)...
Ch. 3.3 - Draw a condensed and a skeletal structure for the...Ch. 3.3 - What is each compounds systematic name?Ch. 3.4 - Prob. 17PCh. 3.4 - Give two names for each of the following alkyl...Ch. 3.5 - Are the following compounds primary, secondary, or...Ch. 3.5 - Name the following amines and tell whether they...Ch. 3.5 - Draw the structures and provide systematic names...Ch. 3.6 - Predict the approximate size of the following bond...Ch. 3.7 - What is the smallest straight-chain alkane that is...Ch. 3.7 - Prob. 24PCh. 3.7 - Prob. 25PCh. 3.7 - Prob. 26PCh. 3.7 - List the compounds in each set from highest...Ch. 3.8 - Rank the following compounds in each set from most...Ch. 3.8 - In which solvent would cyclohexane have the lowest...Ch. 3.8 - Prob. 30PCh. 3.9 - Prob. 31PCh. 3.9 - a. Draw the three staggered conformations and the...Ch. 3.9 - a. Draw the most stable conformation of pentane...Ch. 3.11 - Draw 1,2,3,4,5,6-hexachlorocyclohexane with a. all...Ch. 3.12 - At any one time, would you expect there to be more...Ch. 3.13 - Prob. 36PCh. 3 - a. How many hydrogens does an alkane with 17...Ch. 3 - Prob. 2PCh. 3 - Draw a condensed structure and a skeletal...Ch. 3 - Prob. 39PCh. 3 - Prob. 40PCh. 3 - Which of the following represents a cis isomer?Ch. 3 - a. How many primary carbons does each of the...Ch. 3 - Name the following amines and tell whether they...Ch. 3 - Which of the following conformers of isobutyl...Ch. 3 - What is each compounds name? a. CH3CH2CH2OCH2CH3Ch. 3 - Draw the structural formula for an alkane that has...Ch. 3 - Which has a. the higher boiling point:...Ch. 3 - Ansaid and Motrin belong to the group of drugs...Ch. 3 - A student was given the structural formulas of...Ch. 3 - Which of the following conformers has the highest...Ch. 3 - Prob. 51PCh. 3 - Draw skeletal structures for the following: a....Ch. 3 - Prob. 53PCh. 3 - For rotation about the C-3 8 C-4 bond of...Ch. 3 - Prob. 55PCh. 3 - What is each compounds systematic name?Ch. 3 - Draw the two chair conformers for each of the...Ch. 3 - Draw the nine constitutional isomers with...Ch. 3 - Prob. 59PCh. 3 - Prob. 60PCh. 3 - Prob. 61PCh. 3 - Using Newman projections, draw the most stable...Ch. 3 - For each of the following disubstituted...Ch. 3 - Prob. 64PCh. 3 - Prob. 65PCh. 3 - What is each compounds systematic name?Ch. 3 - Prob. 67PCh. 3 - Bromine is a larger atom than chlorine, but the...Ch. 3 - Prob. 69PCh. 3 - Prob. 70PCh. 3 - a. Draw a potential energy diagram for rotation...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Which of the following solutions has the higher molarity? 10 ppm KI in water or 10,000 ppb KBr in water 0.25 ma...
CHEMISTRY-TEXT
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
Classify each example of molecular art as a pure element, a pure compound, or a mixture.
General, Organic, and Biological Chemistry - 4th edition
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the following compounds in order of increasing melting point.arrow_forwardWhich of the following is least soluble in water? A. CH3OH B. CH3CH2CH2OH C. CH3CH2OH D. CH3CH2CH2CH2OH E. HOCH2OHarrow_forward4-Methylphenol, CH3C6H4OH (pKa 10.26), is only slightly soluble in water, but its sodium salt, CH3C6H4O- Na+, is quite soluble in water. In which solution(s) will 4-methylphenol dissolve? Q.) Aqueous NaOHarrow_forward
- Rank the following compounds in order of increasing solubility in waterarrow_forwardarrange the following compounds in order of increasing boiling point. Circle the mostwater soluble compound.arrow_forwardFor each of the following pairs, predict which substance is more soluble in water. (a) CH3CH2NH2 or NH3 (b) (CH3)3COH or CH3(CH2)5OH (c) CH3CH2CH2OH or CH3CH2CH2CH3 (d) CH3CH2CN or CH3CH2OCH3 (e) CH3CH2OH or CH3CH2CH2OH (f) CH3CH2OCH3 or CH3CH2CO2Harrow_forward
- Match the given compounds with their expected solubility. soluble in 5% HCl soluble in 5% NaOH insoluble in water and concentrated H2SO4 soluble in water a. hexane b. decanoic acid c. methanol d. octanaminearrow_forwardRank the following compounds in order of increasing solubility in hexane:arrow_forwardPredict which member of each pair will be more soluble in water. Explain the reasonsfor your answers. heptan-1-ol or 4-methylphenolarrow_forward
- Rank the attached compounds in order of increasing water solubility ?arrow_forwardPredict which member of each pair will be more soluble in water. Explain the reasonsfor your answers.(a) hexan-1-ol or cyclohexanolarrow_forwardWhich of the following compounds will be most soluble in toluene(C7H8)? A) ethylene glycol (HOCH2CH2OH) B) hexane (CH3CH2CH2CH2CH2CH3) C) acetone (CH3COCH3) D) trimethylamine (N(CH3)3) E) A and D are soluble in toluene.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY