
Interpretation:
The other experiment which dispute of J. J. Thomson’s plum pudding atomic model needs to be explained.
Concept introduction:
In Thomson's plum pudding model, in an atom the electrons are present in a positive charge sphere like seeds in a watermelon. The positive matter was thought to be pulp or a softy material. The total positive charge inside the atom is equal to the total negative charge carried by the electron, this makes the atom electrically neutral.
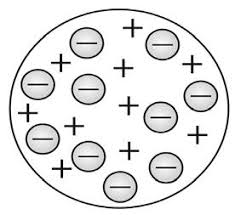
Plum pudding model
Explanation of Solution
The Rutherford modified the atomic model of Sir J. J. Thomson. According to the accepted atomic model, in which an
The Rutherford’s experiment of thin gold foil has different results from the plum pudding model.
The step up is represented as follows:
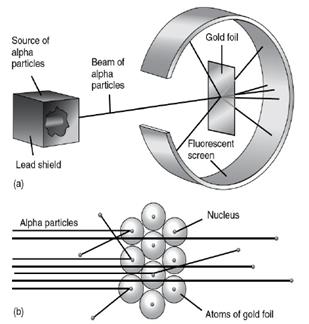
Rutherford model
It was Rutherford's thin gold foil experiment. The result contradicts the plum pudding atomic model.
Chapter 4 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Organic Chemistry
Organic Chemistry (8th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry: Structure and Properties
Organic Chemistry (9th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





