
Write the formulas of the following compounds and decide which are soluble in water.
(a) sodium sulfate
(b) iron(lll) nitrate
(c) silver chloride
(d) chromium(lll) hydroxide
(a)
Interpretation:
The formula for sodium sulfate should be written along with identify whether it is soluble in water or not.
Concept introduction:
First, the symbol of the metal (cation) with its ion charge as a superscript should be written.
The symbol of the non-metal (anion) with its ion charge or polyatomic ion as a superscript should be written.
After that, the charges should be crisscross so that they become subscript for the opposite element and + and - charges should be removed.
| Group-1 cations and |
||||||
| Group-2 cations | - | - | ||||
| Transitions metal cations |
- | - | - |
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with hyphen will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Answer to Problem 1QAP
The formula is
It is soluble in water.
Explanation of Solution
Given compound is sodium sulfate
Symbol of sodium is
Symbol of sulfate is
Sodium belongs to group 1 and carries +1 charge.
Sulfate is a polyatomic ion carries -2 charge.
The symbol of sodium and sulfate is written with their charges and charges are crisscrossed.
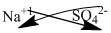
Thus, the formula is
Ions present in the solution is
According to solubility, attractive forces present between ion and ion is less than the water to ion. Thus, it is soluble in water.
(b)
Interpretation:
The formula for iron(III) nitrate should be written along with identify whether it is soluble in water or not.
Concept introduction:
First, the symbol of the metal (cation) with its ion charge as a superscript should be written.
The symbol of the non-metal (anion) with its ion charge or polyatomic ion as a superscript should be written.
After that, the charges should be crisscross so that they become subscript for the opposite element and + and - charges should be removed.
| Group-1 cations and |
||||||
| Group-2 cations | - | - | ||||
| Transitions metal cations |
- | - | - |
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with hyphen will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Answer to Problem 1QAP
The formula is
It is soluble in water.
Explanation of Solution
Given compound is iron(III) nitrate
Symbol of iron is
Symbol of nitrate is
Here, iron(III) implies iron carries +3 charge.
Nitrate is a polyatomic ion carries -1 charge.
The symbol of iron and nitrate is written with their charges and charges are crisscrossed.
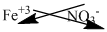
Thus, the formula is
Ions present in the solution is
According to solubility chart, all nitrates are soluble. Thus, iron(III) nitrate is soluble in water.
(c)
Interpretation:
The formula for silver chloride should be written along with identify whether it is soluble in water or not.
Concept introduction:
First, the symbol of the metal (cation) with its ion charge as a superscript should be written.
The symbol of the non-metal (anion) with its ion charge or polyatomic ion as a superscript should be written.
After that, the charges should be crisscross so that they become subscript for the opposite element and + and - charges should be removed.
| Group-1 cations and |
||||||
| Group-2 cations | - | - | ||||
| Transitions metal cations |
- | - | - |
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with hyphen will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Answer to Problem 1QAP
The formula is
It is insoluble in water.
Explanation of Solution
Given compound is silver chloride
Symbol of silver is
Symbol of chloride is
Here, silver is a metal carries + 1 charge.
Chloride is a non-metal carries -1 charge.
The symbol of silver and chloride is written with their charges and charges are crisscrossed.
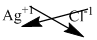
The formula is
Make the above formula simplify, thus the formula of given compound is
Ions present in the solution is
According to the solubility chart, it will form precipitate, thus insoluble in water.
(d)
Interpretation:
The formula for chromium(III) hydroxide should be written along with identify whether it is soluble in water or not.
Concept introduction:
First, the symbol of the metal (cation) with its ion charge as a superscript should be written.
The symbol of the non-metal (anion) with its ion charge or polyatomic ion as a superscript should be written.
After that, the charges should be crisscross so that they become subscript for the opposite element and + and - charges should be removed.
| Group-1 cations and |
||||||
| Group-2 cations | - | - | ||||
| Transitions metal cations |
- | - | - |
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with hyphen will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Answer to Problem 1QAP
The formula is
It is insoluble in water.
Explanation of Solution
Given compound is chromium(III) hydroxide
Symbol of chromium is
Symbol of hydroxide is
Here, chromium(III) implieschromium carries +3 charge.
Hydroxide ion carries -1 charge.
The symbol of chromium and hydroxide is written with their charges and charges are crisscrossed.
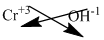
The formula is
Ions present in the solution is
According to the solubility chart, it will form precipitate, thus insoluble in water.
Want to see more full solutions like this?
Chapter 4 Solutions
Chemistry: Principles and Reactions
- ssume a highly magnified view of a solution of HCI that allows you to “see” the HCl. Draw this magnified view. If you dropped in a piece of magnesium, the magnesium would disappear, and hydrogen gas would he released. Represent this change using symbols for the elements, and write the balanced equation.arrow_forwardA 10.0-mL sample of potassium iodide solution was analyzed by adding an excess of silver nitrate solution to produce silver iodide crystals, which were filtered from the solution. KI(aq)+AgNO3(aq)KNO3(aq)+AgI(s) If 2.183 g of silver iodide was obtained, what was the molarity of the original KI solution?arrow_forwardUse the term soluble, insoluble, or immiscible to describe the behavior of the following pairs of substances when they are shaken together: a.25mL of water and 1g of salt the resulting mixture is clear and colorless. b.25mL of water and 1g of solid silver chloride the resulting mixture is cloudy and solid settles out. c.25mL of water and 5mL of mineral oil the resulting mixture is cloudy and gradually separates into two layers.arrow_forward
- A 25.0-mL sample of sodium sulfate solution was analyzed by adding an excess of barium chloride solution to produce barium sulfate crystals, which were filtered from the solution. Na2SO4(aq)+BaCl2(aq)2NaCl(aq)+BaSO4(s) If 5.719 g of barium sulfate was obtained, what was the molarity of the original Na2SO4 solution?arrow_forwardCalcium carbonate, CaCO3, can be obtained in a very pure state. Standard solutions of calcium ion are usually prepared by dissolving calcium carbonate in acid. What mass of CaCO3 should be taken to prepare 500. mL of 0.0200 M calcium ion solution?arrow_forwardWhat is the difference between a solute and a solvent?arrow_forward
- 4-81 (Chemical Connections 4C) Balance the lithium iodine battery redox reaction described in this sec tion and identify the oxidizing and reducing agents present.arrow_forwardWhat is the molarity of pure water with a density of 1.00 g/mL?arrow_forwardBone was dissolved in hydrochloric acid, giving 50.0 mL of solution containing calcium chloride, CaCL2. To precipitate the calcium ion from the resulting solution, an excess of potassium oxalate was added. The precipitate of calcium oxalate, CaC2O4, weighed 1.437 g. What was the molarity of CaCl2 in the solution?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





