
Concept explainers
Two electrostatic potential maps are shown, one of methyllithium (CH3Li) and the other of chloromethane (CH3Cl). Based on their polarity patterns, which do you think is which?


Interpretation:
From given electrostatic potential maps of methyllithium and chloromethane which is which has to be predicted based on polarity pattern.
Concept introduction:
The degree of electron transfer in polar covalent bond can be pictured with something called electrostatic potential maps which uses the colour to represent the electron distribution in an isolated gas-phase molecules.
In the electrostatic potential map colour of the parts are indicates given,
Yellow-green colour - nonpolar or neutral atom
Blue colour - an electron poor atom
Red colour- an electron rich atom.
Answer to Problem 4.26CP
(a) is found as chloromethane and (b) is found as methyllithium.
Explanation of Solution
Given:
Given two electrostatic potential maps are,
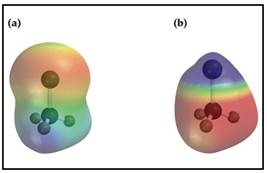
Figure 1
To predict: Methyllithium and chloromethane from given electrostatic potential maps
In electrostatic potential maps Lithium and Chlorine are placed at the top of each map.
Red colour area is for negatively polarized region hence which is accompanying with Chlorine.
The blue colour area is for passively polarized region hence it is accompanied with Lithium.
Thus, map (a) is for Methylchloride
Want to see more full solutions like this?
Chapter 4 Solutions
General Chemistry
- Acrolein is the starting material for certain plastics. (a) Which bonds in the molecule are polar and which are nonpolar? (b) Which is the most polar bond in the molecule? Which atom is the partial negative end of this bond?arrow_forwardDinitrogen difluoride, N₂F₂, is the only stable, simple inor-ganic molecule with an N=N bond. The compound occurs incisand transforms.(a) Draw the molecular shapes of the two forms of N₂F₂.(b) Predict the polarity, if any, of each form.arrow_forwardDraw a three dimensional model of methanol, CH3OH, including lone pairs of electrons using wedges, dashes and straight linesarrow_forward
- Is an ion considered to be polar, even if it does not contain any polar bonds? think about what the electronegative value applies to? Can electronegativity values be applied to atoms in which electrons have been added or removed from their normal Valencia’s?arrow_forwardhat does it mean to say that a bond is polar? Give two examples of molecules with polar bonds. Indicate in your examples the direction of the polarity.arrow_forwardAcrylonitrile is the building block of the synthetic fiber Orlon. Acrylonitrile Which carbon-carbon bond is shorter? Which carbon-carbon bond is stronger? Which bond is the most polar? What is the partial negative end of that bond?arrow_forward
- Which one of the following bonds is polar? a. a bond between two identical atoms b. a bond between two atoms with the same electronegativity c. a bond between two atoms with different electronegativitiesarrow_forwardSuccessive substitution of F atoms for H atoms in the molecule CH4 produces the molecules CH3F, CH2F2, CHF3, and CF4. a. Draw Lewis structures for each of the five molecules. b. Using VSEPR theory, predict the geometry of each of the five molecules. c. Specify the polarity (polar or nonpolar) for each of the five molecules.arrow_forwardWhich molecule has the most polar bond: N2, BrF, or ClF? Use an arrow to show the direction of polarity in each bond.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning





