
CNCT ORG CHEM 6 2020
6th Edition
ISBN: 9781266807244
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 4, Problem 42P
Interpretation Introduction
Interpretation: To determine the increasing order of boiling point of the given
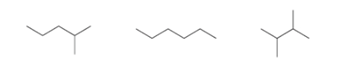
Concept Introduction: Alkane’s boiling point increases with an increase in the carbon chain length. This is because despite being weak individually, the intermolecular attractive forces get cumulatively stronger as the number of carbon atoms increases.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
"A cis alkene is more polar than a trans alkene, giving it a slightlyhigher boiling point and making it more soluble in polar solvents" Explain this statement ?
Give the correct IUPAC name of the following compound:
...
A 7-cyclohexyl-3,5,5-trimethyl-4-pentylheptane
B 1-cyclohexyl-4-sec-butyl-3,3-dimethylnonane
1-cyclohexyl-3,3,5-trimethyl-4-pentylheptane
D 4-sec-butyl-1-cyclohexyl-3,3-dimethylnonane
E none of these
Provide the correct IUPAC name for the skeletal (line-bond) structure
shown here.
6,7-| 6-|| 7- | 4-
3- 2,3-
neo
di
sec-
iso
tert-
eth hex[ but
meth oct non
yl
ane
ene
yne
Chapter 4 Solutions
CNCT ORG CHEM 6 2020
Ch. 4.1 - Prob. 1PCh. 4.1 - Problem 4.2 Which of the following is not another...Ch. 4.1 - Problem 4.3 Draw the five constitutional isomers...Ch. 4.1 - Prob. 4PCh. 4.1 - Prob. 5PCh. 4.2 - Draw the five constitutional isomers that have...Ch. 4.4 - Problem 4.7 Give the IUPAC name for each...Ch. 4.4 - Give the IUPAC name for each compound. a....Ch. 4.4 - Problem 4.9 Give the structure corresponding to...Ch. 4.4 - Prob. 10P
Ch. 4.5 - Give the IUPAC name for each compound.Ch. 4.5 - Give the structure corresponding to each IUPAC...Ch. 4.8 - Arrange the following compounds in order of...Ch. 4.9 - Problem 4.14 Draw the staggered and eclipsed...Ch. 4.9 - Prob. 15PCh. 4.9 - Prob. 16PCh. 4.10 - Problem 4.17 a. Draw the three staggered and...Ch. 4.10 - Problem 4.18 Rank the following conformations in...Ch. 4.10 - Problem 4.19 Consider rotation around the...Ch. 4.10 - Calculate the destabilization present in each...Ch. 4.12 - Problem 4.21 Classify the ring carbons as up or...Ch. 4.12 - Problem 4.22 Using the cyclohexane with the C’s...Ch. 4.13 - Draw a second chair conformation for each...Ch. 4.13 - Problem 4.24 Draw both conformations for and...Ch. 4.13 - Problem 4.25 Draw the structure for each compound...Ch. 4.13 - Prob. 26PCh. 4.14 - Prob. 31PCh. 4.14 - Prob. 32PCh. 4.15 - Prob. 33PCh. 4 - Name each alkane using the ball-and-stick model,...Ch. 4 -
4.40 Draw the structure corresponding to each...Ch. 4 - 4.42 Give the IUPAC name for each compound.
a....Ch. 4 - Prob. 42PCh. 4 - 4.46 Considering rotation around the bond...Ch. 4 - 4.50 Calculate the barrier to rotation for each...Ch. 4 - 4.51 The eclipsed conformation of is less...Ch. 4 - (a) Draw the anti and gauche conformations for...Ch. 4 - For each compound drawn below: a.Label each OH,Br...Ch. 4 - Draw the two possible chair conformations for...Ch. 4 - For each compound drawn below: a. Draw...Ch. 4 - Classify each pair of compounds as constitutional...Ch. 4 - Prob. 66PCh. 4 - 4.64 Draw the products of combustion of each...Ch. 4 - 4.65 Hydrocarbons like benzene are metabolized in...Ch. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Cyclopropane and cyclobutane have similar strain...Ch. 4 - Prob. 72PCh. 4 - Haloethanes (CH3CH2X,X=Cl,Br,I) have similar...Ch. 4 - Prob. 74PCh. 4 - Prob. 75PCh. 4 - Consider the tricyclic structure B (a) Label each...Ch. 4 - Read Appendix B on naming branched alkyl...Ch. 4 - Read Appendix B on naming bicyclic compounds. Then...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which compound would you expect to have the highest boiling point? Ethene Bromoethane Ethane O Ethyne mpound is/are meso:arrow_forwardpravin Rank the following alkanes in order increasing boiling pointarrow_forwardDraw all constitutional isomers formed by monochlorination of each alkane.arrow_forward
- Give the structure corresponding to following name. (R)-1-fluoro-2,6,6-trimethylnonanearrow_forwardCyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?arrow_forwardDraw the structures based on iupac name digitally (highly suggest chem-space .com for easy drawing) 1,1-Dibromo-4-isopropylcyclohexane 4-sec-Butyl-2-chlorononane 1.1-Dibromo-4-Pert-Butylcyclohexanearrow_forward
- Provide the correct IUPAC name for the skeletal (line-bond) structure shown here. 4- 1- 3- 2- 5- 7- iso neo di sec- tert-arrow_forwardExplain Conformations of Acyclic Alkanes—Ethane ?arrow_forwardEssentials of General, Organic, and Biochemistry Number the longest chain of carbon atoms so as the Denise Guinn THIRD EDITION presented by Macmillan Learning substituents have the lowest possible numbers. Carbon Give the IUPAC name of the branched alkane. atoms that are not in the longest chain should not be numbered.arrow_forward
- Draw all alkenes that react with one equivalent of H2 in the presence of a palladium catalyst to form each alkane. Consider constitutional isomers only.arrow_forwardGive the structure corresponding to following IUPAC name. trans-3-methylcyclopentanecarbaldehydearrow_forwardWhich of the following is false in regard to alkanes? Group of answer choices very unreactive unsaturated classified as alphatic none of these come in chains and/or ringsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning