
A dilute aqueous solution of H2SO4 (Solution A) is to be mixed with a solution containing 90.0 wt% H2SO4 (Solution B) to produce a 75.0 wt% solution (Solution C).
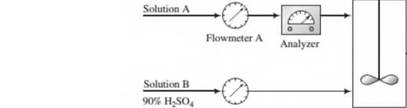
The flow rate and concentration of Solution A change periodically, so that it is necessary to adjust the flow rate of Solution B to keep the product H2SO4 concentration constant.
Flowmeters A and B have linear calibration plots of mass flow rate (m) versus meter reading (/?),
| which pass through the following points: | ||
| Flowmeter A: | mA = 150 lbn,/h, wa = 500 lbm/h. | «a = 25 «a = 70 |
| Flowmeter B: | riiB = 200 lbm/h, mB= 800 lbm/h. | Rb = 20 /?B = 60 |
The analyzer calibration is a straight line on a semilog plot of %H2SO4(x) on a logarithmic scale versus meter reading (/?,) on a linear scale. The line passes through the points (x = 20%, Rx= 4.0) and (x = 100%./?, = 10.0).
- Calculate the flow' rate of Solution B needed to process 300 lbm/h of 55% H2SO4 (Solution A), and the resulting flow rate of Solution C. (The calibration data are not needed for this part.)
- Derive the calibration equations for /ha(#a). «ib(^b), andx(/?v). Calculate the values of R\, Ru, and Rxcorresponding to the flow rates and concentrations of Part (a).
- The process technician's job is to read Flowmeter A and the analyzer periodically, and then to adjust the flow rate of Solution B to its required value. Derive a formula that the technician can use for Rbin terms of RAand Rx, and then check it by substituting the values of Part (a).
Learn your wayIncludes step-by-step video

Chapter 4 Solutions
Elementary Principles of Chemical Processes, Binder Ready Version
Additional Engineering Textbook Solutions
Process Dynamics and Control, 4e
Elements of Chemical Reaction Engineering (5th Edition) (Prentice Hall International Series in the Physical and Chemical Engineering Sciences)
Concepts Of Programming Languages
Management Information Systems: Managing The Digital Firm (16th Edition)
Heating Ventilating and Air Conditioning: Analysis and Design
INTERNATIONAL EDITION---Engineering Mechanics: Statics, 14th edition (SI unit)
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





