
(a)
Interpretation:
Given molecular compound has to be named.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
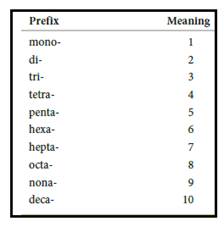
Figure 1
(b)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(c)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
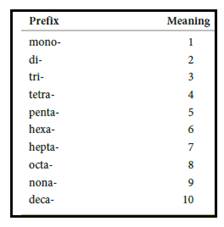
Figure 1
(d)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
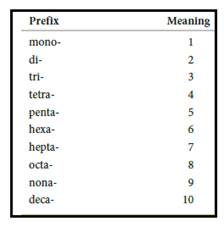
Figure 1
(e)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(f)
Interpretation:
Given molecular compound has to be named.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(g)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General Chemistry: Atoms First
- 2.54 Based on what you have learned in this chapter, would you classify the chemistry of polymers as organic or inorganic? Why?arrow_forward(a) Which two of the following are isotopes of the same element:3116X, 3115X, 3216X? (b) What is the identity of the elementwhose isotopes you have selected?arrow_forwardis the chemical formula, C2OH2N2O2, how would you write 3 molecules of this compoundarrow_forward
- How many hydrogen atoms are in each of the following:(a) C2H5OH, (b) Ca1C2H5COO22, (c) 1NH423PO4?arrow_forwardHexane is C6H14. What is the formula for the alkane with 3 more carbons?arrow_forward31. What is the empirical formula of the compounds with the following molecular formulas? (a) C10H22 (b) N2O5 (c) C5H15O10arrow_forward
- name the following compounds. I need part K, L, Sarrow_forwardConsider a compound with 53% carbon, 36% oxygen and 11% hydrogen. What formula would a compound with this composition have?arrow_forwardWhat is the name of the compound whose formula is (CH3)2AsO2H? A.) Arsenous acid B.) Arsenic acid C.) trimethylarsine oxide D.) monomethylarsonic acid E.) dimethylarsinic acidarrow_forward
- Chemistry A molecular compound has the formula, C6H10. Enter the name of the compound. -Not cyclohexane.arrow_forwardIn the molecule C6H12N6, how many C, H and N atoms are there?arrow_forwardCalculate the molecular mass of the following substances: (a) Ibuprofen, C13H18O2 (a drug used as for pain relief)(b) Phenobarbital, C12H12N2O3 (a drug used as a sedative, hypnotic, and antiseizure medication)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

