
(a)
Interpretation:
Given molecular compound has to be named.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
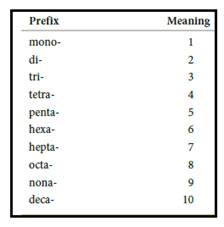
Figure 1
(b)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(c)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
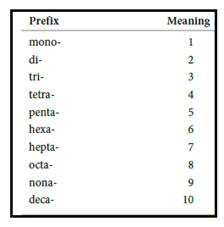
Figure 1
(d)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
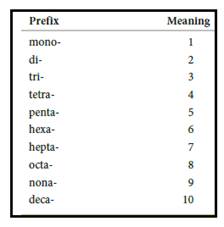
Figure 1
(e)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(f)
Interpretation:
Given molecular compound has to be named.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(g)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(h)
Interpretation:
The systematic name for given compound has to be given.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General Chemistry: Atoms First
- Chemistry A molecular compound has the formula, C6H10. Enter the name of the compound. -Not cyclohexane.arrow_forward31. What is the empirical formula of the compounds with the following molecular formulas? (a) C10H22 (b) N2O5 (c) C5H15O10arrow_forwardGive the Systematic names of the following compounds:d. SO2______________e. N2O2___________f. HI___________g. H2SO4___________arrow_forward
- In the molecule C6H12N6, how many C, H and N atoms are there?arrow_forwardRemdesivir (C27H35N6O8P1) is viewed as one of the most promising treatments for patients suffering from COVID-19 so there are many clinical trails underway. Determine the number of P, N, O, C, and H atoms in 0.500g of C27H35N6O8P1arrow_forwardis the chemical formula, C2OH2N2O2, how would you write 3 molecules of this compoundarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning


