
(a)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
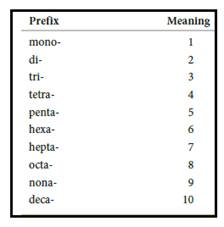
Figure 1
(b)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
(c)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
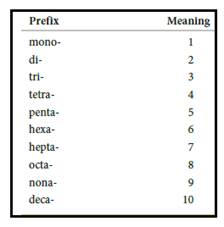
Figure 1
(d)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,
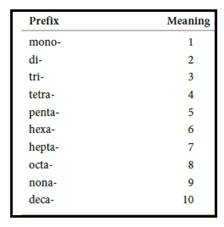
Figure 1
(e)
Interpretation:
For given named compound the formula has to be written.
Concept introduction:
Naming molecular compounds:
To named binary ionic compounds, first predict the positive ion and then negative ion. The positive ion as named as same element while negative ion taking the first part from its original name and ending with –ide.
Numerical prefix for naming compounds are,

Figure 1
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General Chemistry: Atoms First
- 3-58 In Section 2-3B, we saw that there are seven diatomic elements. (a) Draw Lewis structures for each of these diatomic elements. (b) Which diatomic elements are gases at room temperature? Which are liquids? Which are solids?arrow_forward1. C3H8 though covalently bonded cannot be named as tricarbon octahydride. Which of the following premise supports this? O The hydrogen acts as cation. O The compound is not an empirical formula O The name follows ionic compound O The number of atoms need not to be specified O C₂Hs is a hydrocarbon 2. Tritium, 1 3 3/1t, a radioactive isotope of hydrogen, slowly turns into a helium isotope, 2 3 He 3/2He. Which of the following is correct about the two isotopes? O They have the same number of neutral subatomic particles O Their cations are deflected to the same point in a magnetic field whose strength is varied OTheir cations are deflected to the same point in a magnetic field whose strength is not varied O They have the same nuclear charge O They have the same number of charges in subatomic particles.arrow_forwardIs there a charge in H2O?arrow_forward
- Which of the following does not exist as a diatomic molecule? Group of answer choices A. Nitrogen B. Hydrogen C. Chlorine D. Fluorine E. Boronarrow_forwardHow many hydrogen atoms are in each of the following:(a) C2H5OH, (b) Ca(C2H5COO)2, (c) (NH4)3PO4?arrow_forwardGiven the pair of compounds,AlCl3 and CaCl2, which is more ionic and why ?arrow_forward
- What is the name of the compound with the formula FeI3 ?arrow_forwardDichlorobenzene, C6H4Cl2, exists in three forms (isomers)called ortho, meta, and para:arrow_forwardPredict the chemical formula for the ionic compoundformed by (a) Ca2+ and Br-, (b) K+ and CO32-, (c) Al3+ andCH3COO-, (d) NH4+ and SO42-, (e) Mg2+ and PO43-.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





