
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
8th Edition
ISBN: 2818440043505
Author: Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 4.12, Problem 40P
The stereoisomer of cholesterol found in nature is shown here.
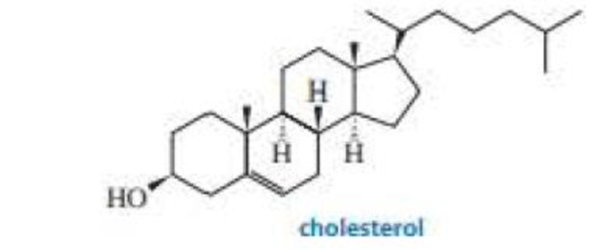
- a. How many asymmetric centers does cholesterol have?
- b. What is the maximum number of stereoisomers that cholesterol can have?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Considerthe image below.
a. Draw the mirror image of structure I and label it as structure II.b. Are I and II chiral?c. What kind of stereocenters do they represent?
The stereoisomer of cholesterol found in nature is shown here.a. How many asymmetric centers does cholesterol have?
b. What is the maximum number of stereoisomers that cholesterol can have?
How many stereoisomers estuary for this molecule? Show how you know.
A. 8
B. 3
C. 4
D. 6
Chapter 4 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
Ch. 4.1 - Prob. 2PCh. 4.1 - Prob. 3PCh. 4.1 - Prob. 4PCh. 4.1 - Prob. 5PCh. 4.1 - Which of the roll owing compounds have a dipole...Ch. 4.2 - Draw and label the E and Z isomers for each of the...Ch. 4.2 - Assign relative priorities to each set of...Ch. 4.2 - Tamoxifen slows the growth of some breast tumors...Ch. 4.2 - Prob. 10PCh. 4.2 - Name each of the following:
Ch. 4.2 - Draw the Z isomer of an alkene that has a CH3 and...Ch. 4.3 - Prob. 13PCh. 4.4 - Prob. 14PCh. 4.5 - Prob. 16PCh. 4.6 - Prob. 17PCh. 4.7 - Prob. 18PCh. 4.8 - Prob. 20PCh. 4.8 - Prob. 22PCh. 4.8 - Prob. 23PCh. 4.8 - Prob. 24PCh. 4.8 - Draw a perspective formula for each or the...Ch. 4.8 - Prob. 27PCh. 4.9 - Prob. 28PCh. 4.9 - What is the configuration of the following...Ch. 4.9 - Prob. 32PCh. 4.10 - Prob. 33PCh. 4.10 - (S)-(+)-Monosodium glutamate (MSG) is a flavor...Ch. 4.11 - Prob. 35PCh. 4.11 - Prob. 36PCh. 4.12 - Prob. 38PCh. 4.12 - Prob. 39PCh. 4.12 - The stereoisomer of cholesterol found in nature is...Ch. 4.12 - Prob. 41PCh. 4.13 - 1-Bromo-2-methylcyclopentane has four pairs of...Ch. 4.13 - Prob. 43PCh. 4.13 - Draw all possible stereoisomers for each of the...Ch. 4.13 - Prob. 45PCh. 4.13 - Of all the possible cyclooctanes that have one...Ch. 4.13 - Prob. 47PCh. 4.13 - Prob. 48PCh. 4.14 - Which of the following compounds has a...Ch. 4.14 - Draw all the stereoisomers for each of the...Ch. 4.15 - Prob. 52PCh. 4.15 - Name the isomers you drew in Problem 52.Ch. 4.15 - Chloramphenicol is a broad-spectrum antibiotic...Ch. 4.15 - Draw a perspective formula for each of the...Ch. 4.15 - Name the following:Ch. 4.15 - Prob. 57PCh. 4.15 - Prob. 59PCh. 4.15 - Convert the perspective formula to a skeletal...Ch. 4.15 - Prob. 62PCh. 4.16 - Prob. 63PCh. 4.17 - Limonene exists as two different stereoisomers....Ch. 4 - a. Draw three constitutional isomers with...Ch. 4 - Prob. 65PCh. 4 - Prob. 66PCh. 4 - Which of the following has an asymmetric center?...Ch. 4 - Prob. 68PCh. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Prob. 71PCh. 4 - Assign relative priorities to each set of...Ch. 4 - Prob. 73PCh. 4 - Which of the following are optically active?Ch. 4 - Prob. 75PCh. 4 - Name the following:Ch. 4 - Which of the following has an achiral...Ch. 4 - Using skeletal structures, draw the stereoisomers...Ch. 4 - Prob. 79PCh. 4 - Citrate synthase, one of the enzymes in the series...Ch. 4 - Prob. 81PCh. 4 - Prob. 82PCh. 4 - Prob. 83PCh. 4 - Prob. 84PCh. 4 - Prob. 85PCh. 4 - Prob. 86PCh. 4 - Prob. 87PCh. 4 - Prob. 88PCh. 4 - Prob. 89PCh. 4 - a. Draw all the isomers with molecular formula...Ch. 4 - Prob. 91PCh. 4 - Prob. 92PCh. 4 - Draw structures for the following: a....Ch. 4 - For each of the following structures, draw the...Ch. 4 - Prob. 95PCh. 4 - Prob. 96PCh. 4 - Prob. 97PCh. 4 - a. Using the wedge-and-dash notation, draw the...Ch. 4 - Prob. 99PCh. 4 - Prob. 100PCh. 4 - Prob. 101PCh. 4 - a. Draw the two chair conformers for each of the...Ch. 4 - Prob. 103PCh. 4 - Is the following compound optically active?Ch. 4 - Prob. 105P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Part D. Do the two structures A and B of each pair drawn below represent the same molecule, constitutional isomers, or stereoisomers? If A and B are stereoisomers, further classify them as enantiomers or diastereomers.arrow_forwardWhich of the following pairs of structures represent a pair of enantiomers? a. b. d. H₂C H NC H₂CH₂C Br HC. H₂N H.... CN COOH Br CH3 CH3 OH COOH H... Br H... HOOC H₂C CN OH HC₂ H₂C H₂N Br CH3 CN CH₂CH3 COOH CHarrow_forwardConsider molecules with more than one chiral center. Consider 2,3-dyhdroxybutanoic acid. a. How many chiral centers does it have? b. Which ones? c. Reproduce the structure and encircle the four groups, which are linked to each chiral carbon.arrow_forward
- why is the stereocenter on the left R?arrow_forwardIn the previous problem, you saw that there were two chiral carbons in 4-bromo-3-hydroxypentanal. That molecule is drawn below with the chiral carbons circled. What is the maximum number of stereoisomers that can exist for this molecule? Can you help me, please? Can you explain to me?arrow_forwardIdentify the configuration for the indicated chiral centers.,To preview image click here A. B. C. HO CI CI a. a. OH Br S HIN b. יוו Br - b. CI b. H Bronchodilator and decongestant Hc. H N Antitumor agent H Vitamin B7arrow_forward
- 1. An object is chiral if it A. is superimposable on its mirror image B. is not superimposable on its mirror image C. has no mirror image D. is identical to each mirror image of itselfarrow_forwardDo you expect each boxed molecule to be chiral or not?arrow_forward9.a. Circle all the appropriate terms. t-Bu The two molecules drawn above are enantiomers optically active enantiomers superimposable 9.b. Circle all the appropriate terms. diastereomers not optically active H optically active Bull.... H₂CH... Br -CH3 The two molecules drawn above are Me H diastereomers not optically active superimposable Me chiral meso not superimposable H3C- H ....... chiral meso H ...III\CH3 Br not superimposable Cl achiral -t-Bu identical achiral identical 3arrow_forward
- a. b. For the following two pairs of molecules, (1) Draw out the chair conformation for each molecule, flip the ring if it is possible. (2) Compare both molecules to circle out which one is more stable. (3) Identify their relationship as: constitutional isomer, conformational isomer, stereoisomer or identical. (4) Find all the chiral center on each molecule and label them. Br.. Ax < and and H. H H Br H Harrow_forwardExamine the models for the presence of a plane of symmetry. Present or Not present? NH2 CH, CH3 CH, H;C -CH3 H,N NH2 H,C--NH, H,C- OH CH, PH2 PH2 CH, C. D. Which are superimposable on their mirror image? A, B, Cand/or D? Which model represents a chiral molecule?A. B. Cond/or D?arrow_forwardIn the answer box, type the number of saymmetric centers in a following molecule. OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
Group Theory - Learn like Expert with 3D animation | Introduction for Beginners | ONE Chemistry; Author: One Chemistry;https://www.youtube.com/watch?v=Lz2ih8fkgDs;License: Standard YouTube License, CC-BY