
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.12, Problem 42P
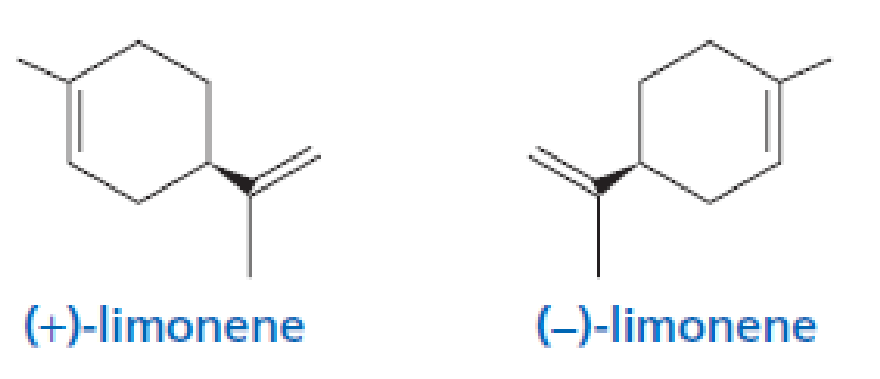
Limonene exists as two different stereoisomers. The R enantiomer is found in oranges and lemons, and the S enantiomer is found in spruce trees. Which of the following is found in oranges and lemons?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Limonene exists as two different stereoisomers. The R enantiomer is found in oranges and lemons, and the S enantiomer is found in spruce trees. Which of the following is found in oranges and lemons?
The structure of 4 isomers of an aldotetrose carbohydrate are given.
1) select every structure that is a diastereomer of structure D
A, B, or C?
2) select every structure that is a enantiomer of structure C
D, B, or A?
3) select every structure that is a stereoisomer of structure D
A, B, or C
Which double bonds in the attached natural products can exhibit stereoisomerism? Nerolidol is isolated from the angel's trumpet plant,caryophyllene is present in hemp, and humulene comes from hops.
Chapter 4 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 4.1 - Draw the cis and trans isomers for the following:...Ch. 4.1 - Prob. 4PCh. 4.1 - Prob. 5PCh. 4.1 - Prob. 6PCh. 4.2 - Prob. 7PCh. 4.2 - Tamoxifen slows the growth of some breast tumors...Ch. 4.2 - Draw and label the E and Z isomers for each of the...Ch. 4.2 - Prob. 10PCh. 4.2 - Name each of the following:Ch. 4.2 - Draw the structure of (Z)-2,3-dimethyl-3-heptene.
Ch. 4.3 - Prob. 13PCh. 4.4 - Prob. 14PCh. 4.5 - Prob. 16PCh. 4.6 - Prob. 17PCh. 4.7 - Assign relative priorities to the groups or atoms...Ch. 4.7 - Name the following:Ch. 4.7 - Prob. 22PCh. 4.7 - Draw a perspective formula for each of the...Ch. 4.8 - Prob. 24PCh. 4.8 - Prob. 27PCh. 4.9 - Prob. 28PCh. 4.9 - Prob. 29PCh. 4.9 - Prob. 30PCh. 4.10 - Prob. 31PCh. 4.10 - Prob. 32PCh. 4.10 - Prob. 33PCh. 4.11 - Prob. 34PCh. 4.11 - a. Draw the stereoisomers of...Ch. 4.11 - Prob. 37PCh. 4.11 - Prob. 38PCh. 4.12 - Which of the following compounds has a...Ch. 4.12 - Draw all the stereoisomers for each of the...Ch. 4.12 - Limonene exists as two different stereoisomers....Ch. 4 - a. Draw three constitutional isomers with...Ch. 4 - Which of the following have an asymmetric center?...Ch. 4 - Prob. 45PCh. 4 - Prob. 46PCh. 4 - Of all the possible cyclooctanes that have one...Ch. 4 - Prob. 48PCh. 4 - Prob. 49PCh. 4 - Prob. 50PCh. 4 - Prob. 51PCh. 4 - Prob. 52PCh. 4 - Draw the stereoisomers of 2,4-dichlorohexane....Ch. 4 - Prob. 54PCh. 4 - Prob. 55PCh. 4 - Prob. 56PCh. 4 - Prob. 57PCh. 4 - Prob. 58PCh. 4 - Prob. 59PCh. 4 - Prob. 60PCh. 4 - Prob. 61PCh. 4 - Prob. 62PCh. 4 - Draw structures for each of the following: a....Ch. 4 - Prob. 64PCh. 4 - a. Draw all the isomers with molecular formula...Ch. 4 - Prob. 66PCh. 4 - Prob. 67PCh. 4 - Prob. 68PCh. 4 - Chloramphenicol is a broad-spectrum antibiotic...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Write a Lewis formula for each of the following organic molecules: C2H3Cl (vinyl chloride: starting material fo...
Organic Chemistry - Standalone book
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Calculate the lattice energy of CaCl2 using a Born-Haber cycle and data from Appendices F and L and Table 7.5. ...
Chemistry & Chemical Reactivity
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- a. What hydrocarbon with molecular formula C4H10 forms only two monochlorinated products? Both products are achiral. b. What hydrocarbon with the same molecular formula as in part a forms three monochlorinated products? One is achiral and two are chiral.arrow_forwardConsider the compound below. a) Draw the structure showing stereochemistry, in which carbon 1 has S configuration and carbon 2 has R configuration. b) Draw the structure showing stereochemistry, in which carbons 1 and 2 have S configuration. c) are the two structures from part a and b diastereomers, identical, enantiomers, or unrelated?arrow_forwardOnly three stereoisomers are possible for 2,3-dibromo-2, 3-dichlorobutane. Draw them, indicating which pair are enantiomers (optical isomers). Why does the other isomer not have an enantiomer?arrow_forward
- Which of the following statement is incorrect? Question 8 options: Achiral molecules having plane of symmetry & 50:50 racemic mixture are optically active Achiral molecules having a plane of symmetry are optically inactive methylcyclohexane has two enantiomers (R and S) Chiral molecules consisting of a single enantiomer are optically active 50:50 Racemic mixture is optically inactivearrow_forwardStereochemistry in biological molecules is often denoted by the D‑ and L‑ convention, instead of the ?‑R‑ and ?‑S‑ configurations determined by the Cahn–Ingold–Prelog methodology. Historically, the glyceraldehyde enantiomer that rotated plane polarized light clockwise was arbitrarily designated as D and the other enantiomer was designated as the L configuration. The D‑glyceraldehyde Fischer projection is shown. D‑glyceraldehydearrow_forwardClassify the following pairs of compounds into a) Connection isomers, b) enantiomers, c) diasteremers, d) the same compound, e) conformations of the same compound.arrow_forward
- Draw structural formulas for the cis and trans isomers of hydrindane. Show each ring in its most stable conformation. Which of these isomers is more stable?arrow_forwardHow many possible stereoisomers?arrow_forwardWhat is the ratio of concentrations of the two conformations at 25 °C? (Calculate the ratio as the conformation present in a higher concentration over the lower one.)arrow_forward
- How stereoisomers differ in configuration ?arrow_forwardsort the compounds into pairs of constitutional isomers, stereoisomers, or identical moleculesarrow_forwardIdentify the stereogenic carbon in (S)- and (R)-limonene, rank the substituents around it and rationalize the assignment of their stereochemical configurations. Hint: When ranking carbons that have multiple bonds, consider the bolded carbon of C=C being connected to 2 carbons and the bolded carbon of C≡C being connected to 3 carbons.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License