
Concept explainers
To find:
(a) in which square in Fig 42.6 of the textbook does the radionuclide 196I transform when it decays by an electron.
(b) if any further decays occur, by looking at Fig 42.6 of the textbook.
Answer to Problem 1Q
Solution:
(a) When the radionuclide 196I decays by an electron, it transforms into 196Pt.
(b) No further decay occurs after 196I decays by an electron, and transforms into 196Pt, because 196Pt is a stable nucleus.
Explanation of Solution
Given:
i. A radionuclide 196I decays by emitting an electron.
ii. Refer to Fig 42.6 of the textbook to know how this nuclide is transformed.
Concept
When a nuclide decays via an electron, a neutron is changing or transforming into a proton, hence the new nuclide will have the same mass A, but a higher Z, that is Z will be transformed to Z+1.
![]()
![]() p + e - +
p + e - +
From Fig 42.6 of the textbook, we get
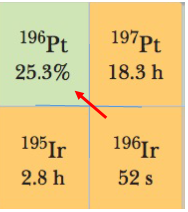
The Z of Ir is 77, and Z of Pt is 78; when 196I decays by emitting an electron, we get
![]()
From Fig 42.6 of the textbook, we can say that
Conclusion
When a nuclide decays by an electron (β- - decay), its Z transforms to Z+1, but it’s mass A remains unchanged.
Want to see more full solutions like this?
Chapter 42 Solutions
Fundamentals of Physics
- Large amounts of 65Zn are produced in copper exposed to accelerator beams. While machining contaminated copper, a physicist ingests 50.0 Ci of 65Zn. Each 65Zn decay emits an average ray energy of 0.550 MeV, 40.0% of which is absorbed in the scientist’s 75.0kg body. What dose in mSv is caused by this in one day?arrow_forward(a) Calculate the energy released in the neutroninduced fission (similar to the spontaneous fission in Example 32.3) n+238U96Sr+140Xe+3n, given m(96Sr)=95.921750u and m(140Xe)=139.92164. (b) This result is about 6 MeV greater than the result for spontaneous fission. Why? (c) Con?rm that the total number at nucleons and total charge are conserved in this reaction.arrow_forward(a) Write the complete a decay equation for 249Cf. (b) Find the energy released in the decay.arrow_forward
- (a) How many 239Pu nuclei must fission to produce a 20.0kT yield, assuming 200 MeV per fission? (b) What is the mass of this much 239Pu?arrow_forward(a) Find the total energy released in MeV in each carbon cycle (elaborated in the above problem) including the annihilation energy. (b) How does this compare with the protonproton cycle output?arrow_forward(a) Calculate the energy released in the neutroninduced fission reaction n+239Pu96Sr+140Ba+4n, given m(96Sr)=95.921750u and m(140Ba)=139.910581u. (b) Confirm that the total number of nucleons and total charge are conserved in this reaction.arrow_forward
- Tritium is naturally rare, but can be produced by the reaction n+2H3H+. How much energy in MeV is released in this neutron capture?arrow_forward(a) Calculate the energy released in the neutron- Induced fission reaction n+235U92Kr+142Ba+2n , given m(92Kr) = 91.926269 u and m(142Ba)= 141.916361 u. (b) Confirm that the total number of nucleons and total charge are conserved in this reaction.arrow_forwardConfirm that charge, electron family number, and the total number of nucleons are all conserved by the rule for (decay given in the equation ZAXNZ+1AYN1++ve. To do this, identify the values of each before and after the decay.arrow_forward
- Confirm That charge, electron family number, and the total number at nucleons are all conserved by the rule for a decay given in the equation ZAXNZ2A4YN2+24He2. To do this, identity the values of each before and after the decay.arrow_forward(a) A cancer patient is exposed to rays from a 5000Ci 60Co transillumination unit for 32.0 s. The rays are collimated in such a manner that only 1.00% of them strike the patient. Of those, 20.0% are absorbed in a tumor having a mass of 1.50 kg. What is the dose in rem to the tumor, it the average energy per decay is 1.25 MeV? None of the s from the decay reach the patient. (b) Is the dose consistent with stated therapeutic doses?arrow_forwardIn the following eight problems, write the complete decay equation for the given nuclide in the complete XZAN notation. Refer to the periodic table for values of Z. decay of 40K, a naturally occurring rare isotope of potassium responsible for some of our exposure to background radiation.arrow_forward
 College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax





