
Interpretation:
The correct orbital diagram for beryllium element needs to be determined.
Concept introduction:
According to the Aufbau principle in the ground state, an electron first enters the lowest energy level and further electrons are added to the orbitals with increasing energies.
Answer to Problem 17STP
Correct answer: The correct option is B.
Explanation of Solution
Reason for correct option:
According to the Aufbau principle, the electron first enters into the lowest energy level. The options are as follows:
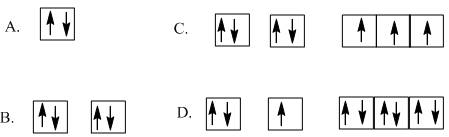
Beryllium contains four electrons and its electronic configuration is
Therefore, the diagram that contains total four electrons is option B.
Reason for incorrect options:
- The total number of electrons is only 2 but in beryllium the number of electrons is 4 thus, option A is incorrect.
C. The total number of valence electrons is 7 but in beryllium the number of electrons is 4 thus, option C is incorrect.
D. The given electronic configuration cannot be true for any element as it does not follow Aufbau principle. Thus, option D is incorrect.
Chapter 5 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: Structure and Properties (2nd Edition)
Organic Chemistry
Chemistry: A Molecular Approach
Organic Chemistry (9th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Organic Chemistry (8th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





