
Concept explainers
Biofuels
A lot of energy is locked up in the
Corn, soy, sugarcane, and other food crops are rich in oils, starches, and sugars that can be easily converted to biofuels. The starch in corn kernels, for example, can be enzymatically broken down to glucose, which is fermented to ethanol by bacteria or yeast. However, growing food crops for biofuel production typically requires a lot of energy (in the form of fossil fuels) and it damages the environment. Making biofuels from other plant matter such as weeds or agricultural waste requires additional steps, because these materials contain a higher proportion of cellulose. Breaking down this tough carbohydrate to its glucose monomers adds cost to the biofuel product.
In 2006, David Tilman and his colleagues published the results of a 10-year study comparing the net energy output of various biofuels. The researchers made biofuel from a mixture of native perennial grasses grown without irrigation, fertilizer, pesticides, or herbicides, in sandy soil that was so depleted by intensive agriculture that it had been abandoned. The energy content of this biofuel and the energy it took to produce it were measured and compared with that of biofuels made from food crops (Figure 5.16).
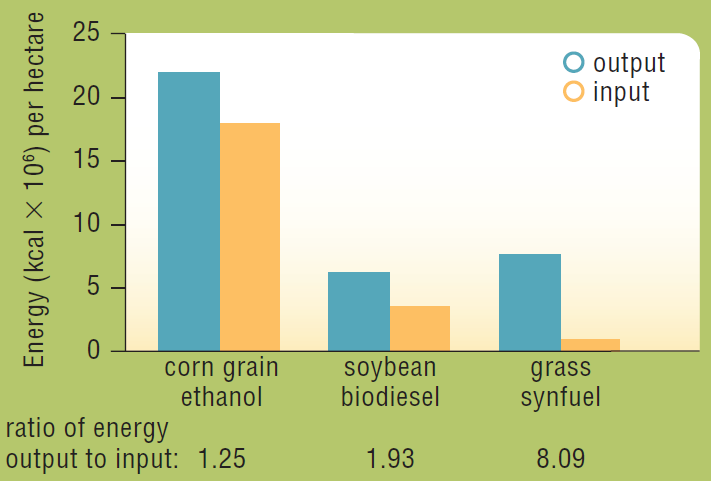
The production of which biofuel was most efficient (which had the highest ratio of energy output to energy input)?
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
EBK BIOLOGY TODAY AND TOMORROW WITHOUT
- Biofuels A lot of energy is locked up in the chemical bonds of molecules made by plants. That energy can fuel consumers, as when an animal cell powers ATP synthesis by aerobic respiration. It can also fuel our cars, which run on energy released by burning biofuels or fossil fuels. Both processes are fundamentally the same: They release energy by breaking the bonds of organic molecules. Both use oxygen to break those bonds, and both produce carbon dioxide. Unlike fossil fuels, biofuels are a renewable source of energy: We can always make more of them simply by growing more plants. Also unlike fossil fuels, biofuels do not contribute to global climate change, because growing plant matter for fuel recycles carbon that is already in the atmosphere. Corn, soy, sugarcane, and other food crops are rich in oils, starches, and sugars that can be easily converted to biofuels. The starch in corn kernels, for example, can be enzymatically broken down to glucose, which is fermented to ethanol by bacteria or yeast. However, growing food crops for biofuel production typically requires a lot of energy (in the form of fossil fuels) and it damages the environment. Making biofuels from other plant matter such as weeds or agricultural waste requires additional steps, because these materials contain a higher proportion of cellulose. Breaking down this tough carbohydrate to its glucose monomers adds cost to the biofuel product. In 2006, David Tilman and his colleagues published the results of a 10-year study comparing the net energy output of various biofuels. The researchers made biofuel from a mixture of native perennial grasses grown without irrigation, fertilizer, pesticides, or herbicides, in sandy soil that was so depleted by intensive agriculture that it had been abandoned. The energy content of this biofuel and the energy it took to produce it were measured and compared with that of biofuels made from food crops (Figure 5.16). About how much energy did ethanol produced from one hectare of corn yield? How much energy did it take to grow and produce that ethanol?arrow_forwardProvide one way you can reduce carbon emissions.arrow_forwardFigure 8.18 Which of the following statements is true? In photosynthesis, oxygen, carbon dioxide, ATP, and NADPH are reactants. G3P and water are products. In photosynthesis, chlorophyll, water, and carbon dioxide are reactants. G3P and oxygen are products. In photosynthesis, water, carbon dioxide, ATP, and NADPH are reactants. RuBP and oxygen are products. In photosynthesis, water and carbon dioxide are reactants. G3P and oxygen are products.arrow_forward
- Which of the following is not an example of an energy transformation? a. Heating up dinner in a microwave b. Solar panels a work c. Formation of static electricity d. None of the abovearrow_forwardAs the sun shines on Earth, plants use its energy to complete the process of photosynthesis. Energy is transformed during the process into chemical energy in the form of sugars, such as carbohydrates. However, the energy available at top consumers is much less than that produced by autotrophs. Why is this so?arrow_forwardWhy do the electrons in water and carbon dioxide have so little chemical energy? Because there are oxygen atoms in these molecules. The high electronegativity of oxygen pulls the electrons to low energy levels. Because they are the end products of cellular respiration and this is the only place where these molecules are produced. Because animals and plants release carbon dioxide and water into the atmosphere. All of the abovearrow_forward
- Why are the rates of photosynthesis and cellular respiration dependent upon temperature?arrow_forwardProvide the balanced chemical reaction for cellular respiration.arrow_forwardThe words exergonic and endergonic can be used to describe many reactions including those essential to life. Photosynthesis is an _reactionarrow_forward
- Your body warms up when you exercise. Why does this happen? Hint: Think back to when we were talking about energy pyramids and the idea that only 10% of energy gets passed up to the next level. What form is the energy that gets lost during inefficient chemical reactions? Нeat CeH1206 Sunlight 02 ATP Cellular Photosynthesis respiration CO2 H,0 + Aa Ctv MacBook Air 888 F10 80 FB FS F4 F3 F2 * & @ # $ % 7 2. 3 4 5 E R T. Y [ W J L G S C V command option and .. .- V - 00 Barrow_forward1. Carbon dioxide +Water+ Energy → Sugar + Oxygen 2. Sugar + Oxygen → Water + Carbon Dioxide + Energy the second reaction shown here (cellular respiration), which of these descriptions compares the products and reactants The process produces ADP as a product, which has a lower energy level compared to the reactants. The process produces inorganic molecules only, which have a lower energy level compared to the products. The process produces ATP as a product, which provides a greater cellular energy compared to the reactants. The process produces heat energy as a byproduct, which causes the reactants to be less energetic compared to the products. Carbon Dloxide Production in Snails & Elodea Test Tube Contents Tube A: control Initial Color Light Set Up Dark Set Up green green blue green green areonarrow_forwardWhat are three ways that carbon is released into the atmosphere?arrow_forward
 Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology Today and Tomorrow without Physiology (Mi...BiologyISBN:9781305117396Author:Cecie Starr, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College



