
Concept explainers
The following four structures are naturally occurring optically active compounds. Star (*) the asymmetric carbon atoms in these structures.
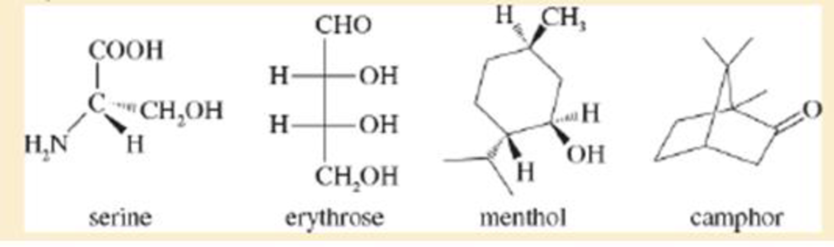
Interpretation: The asymmetric carbon atoms in the given structures are to be marked by star.
Concept introduction: A chiral carbon atom is attached to four different atoms or group of atoms and shows a tetrahedral geometry. The mirror image of a chiral compound is non-super imposable. The two different forms in which a single chiral carbon can exist are referred as enantiomers. The number of enantiomers of a molecule depends on the number of chiral centers.
To determine: The asymmetric carbon atoms in the given structures marked by star.
Answer to Problem 5.25SP
The asymmetric carbon atoms in the given structure have been marked by star.
Explanation of Solution
The given compound is serine. It is attached to
The asymmetric carbon atom present in it is marked by star as shown below.
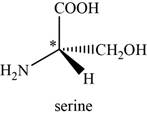
Figure 1
The given compound is erythrose. There are two chiral centres present in it.
The asymmetric carbon atoms present in it are marked by star as shown below.
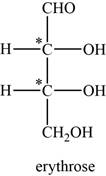
Figure 2
The given compound is menthol. There are three chiral centres present in it.
The asymmetric carbon atoms present in it are marked by star as shown below.
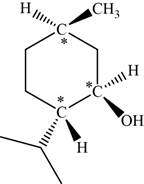
Figure 3
The given compound is camphor. There are two chiral centres present in it.
The asymmetric carbon atoms present in it are marked by star as shown below.
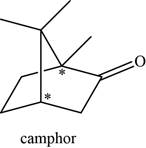
Figure 4
The asymmetric carbon atoms in the given structure have been marked by star.
Want to see more full solutions like this?
Chapter 5 Solutions
ORGANIC CHEMISTRY
Additional Science Textbook Solutions
Chemistry For Changing Times (14th Edition)
Chemistry: Structure and Properties (2nd Edition)
Chemistry (7th Edition)
General, Organic, and Biological Chemistry (3rd Edition)
Chemistry In Context
General Chemistry: Principles and Modern Applications (11th Edition)
- Draw all possible isomers of trimethylcyclopropane. Name them. Which of these isomers are chiral? Indicate by underlining the name(s).arrow_forwardExplain What are conformational isomerism?arrow_forwardThe energy difference between a tert-butyl group going from equatorial to axial in a cyclohexane is 18.3 kJ/mol. When two of the carbon atoms are replaced with oxygen atoms (molecule B) the energy difference between the two chair conformations drops to 5.9 kJ/mol. Explain this difference. (Hint: Consider what makes putting groups axial unfavorable).arrow_forward
- A student drew a noncyclic compound that has the molecular formula C7H14.This molecular formula is representative of an alkane, alkene, alkyne or aromatic and would be classified as saturated or unsaturated ?arrow_forwardIllustrate the Biologically active organic compounds that contain conjugateddouble bonds ?arrow_forward5a) Construct a model of butene, C4H8, using two carbon atoms with planar geometry and two carbon atoms with tetrahedral geometry. How many other isomers of C₂H, can you construct? Draw a dash structural representation of each structure. 5b) How many bonds in butene enjoy free rotation?arrow_forward
- Draw the skeletal (line-bond) structure of [CH3CH2CH2CH2O]-. Include all lone pairs and charges as appropriate.arrow_forwardAnswer this question:How to draw the line bond formula or lewis structure of a methyl ketone with the chemical formula C6H5C3H5O? (also taking into consideration the solubility test and chemical test results provided below) Based on the results of the solubility tests the compound is insoluble in water, 10% NaOH and 10% HCl but soluble in concentrated H2SO4. The functional group/class is identified to be Methyl Ketone, based on the results of the chemical tests on Table 2. CHEMICAL TEST OBSERVATIONS +(compound tested positive for the chemical reaction)/ otherwise (-) Molisch test turbid colorless solution - 2,4-DNP test formation of orange-yellow precipitates + Tollen’s test turbid colorless solution - Ninhydrin test clear pale-yellow solution - iodoform test clear pale-yellow solution +arrow_forward1. Although there is only one alkene with the formula C2H4 (ethene) and only one with the formula C3H6 (propene), there are several alkenes with the formula C4H8. Draw all of the possible bond line structures for alkenes with the formula C4H3 including all possible structural and stereoisomers.arrow_forward
- 5 . Term describing the structural relationship between (2R,3R,4S)-2,3,4-trichloroheptane and (2R,3R,4R)-2,3,4-trichloroheptanearrow_forwardHow many structural isomers of the molecular formula C₃H₆O do not have cyclic structures? Hint: You need to consider placing more than one functional group in a molecule.arrow_forwardDefine Spectroscopic Properties of of Alkanes ?arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





