
Concept explainers
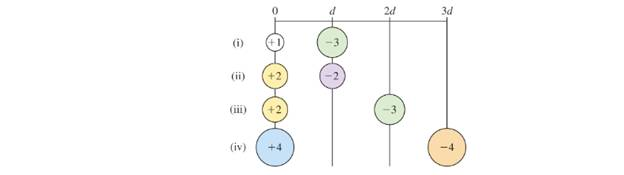
Arrange the following pairs of charged particles in order of increasing electrostatic potential energy.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Student Solutions Manual for Chemistry
Additional Science Textbook Solutions
EBK INTRODUCTION TO CHEMISTRY
Chemistry
Introduction to Chemistry
Chemistry: The Central Science (13th Edition)
Organic Chemistry - Standalone book
Thermodynamics, Statistical Thermodynamics, & Kinetics
- Calculate Kb for each of the following ions: CN-, F-, CH3COO-, HCO3arrow_forwardAt large interatomic separations, an alkali halide molecule MX has a lower energy as two neutral atoms, M+X ; at short separations, the ionic form (M+)(X) has a lower energy. At a certain distance, Rc , the energies of the two forms become equal, and it is near this distance that the electron will jump from the metal to the halogen atom during a collision. Because the forces between neutral atoms are weak at large distances, a reasonably good approximation can be made by ignoring any variation in potential V(R) for the neutral atoms between Rc and R= . For the ions in this distance range, Rc is dominated by their Coulomb attraction. (a) Express Rc for the first ionization energy of the metal M and the electron affinity of the halogen X. (b) Calculate Rc for LiF, KBr, and NaCl using data fromAppendix F.arrow_forwardUse the data in Table 3.1 to plot the logarithm of ionization energy versus the number of electrons removed for Be. Describe the electronic structure of the Be atom.arrow_forward
- Schrodinger and de Broglie suggested a ‘Wave—particle duality" for small particles—that is, if electromagnetic radiation showed some particle-like properties, then perhaps small punicles might exhibit same wave-like properties. Explain. How does the wave mechanical picture of the atom fundamentally differ from the Bohr model? How do wave mechanical arbitals differ from Bohr’s orbits? What does it mean to say that an orbital represents a probability map for an electron?arrow_forwardUse the data in Figure 3.11 and Table 3.2 to calculate the energy changes (E) for the following pairs of reactions: (a) Na(g)+I(g)Na+(g)+I(g) Na(g)+I(g)Na(g)+I+(g) (b) Rb(g)+Br(g)Rb+(g)+Br(g) Rb(g)+Br(g)Rb(g)+Br+(g) Explain why Na+I and Rb+Br form in preference to NaI+ and RbBr+ .arrow_forwardWithout consulting any tables, arrange the following substances in order and explain your choice of order: (a) Mg2+,Ar,Br,Ca2+ in order of increasing radius (b) Na, Na+ , O, Ne in order of increasing ionization energy (c) H, F, Al, O in order of increasing electronegativityarrow_forward
- help pls is this answer corrcet, i cant stack li+ and cu- on top of each other ?arrow_forwardA positively charged particle and a negatively charged particleare initially far apart. What happens to their electrostaticpotential energy as they are brought closer together?arrow_forwardIf the ions in the figure on the right were released and allowedto move, would they move closer together or farther apart?arrow_forward
- Which have highest lattic energy? Explain the answer!arrow_forwardCan you please exlplain how you know how many electrons are present in the pi system??arrow_forwardIn your own words, explain the meaning of “Conjugation” and give anexample of its use with a figure to explain your examplearrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


