
(a)
Interpretation:
The major product for reaction between
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Reduction Reaction: It is just opposite of oxidation reaction which involves removal of oxygen atoms or addition of hydrogen atoms and addition of electrons.
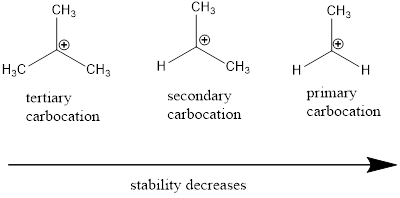
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
(b)
Interpretation:
The major product for reaction between
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Reduction Reaction: It is just opposite of oxidation reaction which involves removal of oxygen atoms or addition of hydrogen atoms and addition of electrons.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

(c)
Interpretation:
The major product for reaction between
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
Addition of halogen to an
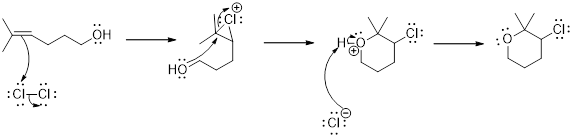
(d)
Interpretation:
The major product for reaction between
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Electrophilic addition: It is a type of addition reaction in which the pi bond present in the molecule breaks as the electrophile approaches and results in the formation of product with sigma bond.
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Acid Catalyzed Hydration Reaction: The reaction involves breaking of phi bonds between carbon-carbon multiple bonds and addition of alcohol to more substituted position of carbon in the molecule.
First step is the acid donates proton to the alkene which leads to the formation of more stable carbo cation.
Then, the water is added to the given alkene through acid catalyzed reaction where the water gets added to the carbo cation finally, the removal of one proton from oxonium ion (oxygen with one positive charge) using water results in the formation of product.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry, Global Edition
- What is the reaction when trans-2-butene reacts with 1.KMnO4/-OH in cold 2. hotKMnO4/-OH 3.HBr in the presence of peroxidearrow_forwardwhat is the compound that most easily reacts with bromine in addition? a) CH4 b) C2H2 c) C3H8 d) C4H10arrow_forwardA haloalkane reacts with a strong base to yield only one alkene product. Select the possible structures that can give this result from Figure 9. * A B C Darrow_forward
- Draw the structure of the minor product formed in the reaction of 2-methylbut-1-ene with concentrated sulfuric acid Explain why thid is the major product? Structure of minor product? Explanation?arrow_forwardDraw the product or products that will be obtained from the reaction of cis-2-butene and trans-2-butene with each of the following reagents. If a product can exist as stereoisomers, show which stereoisomers are formed. 1. HCl. 4.Br2 in CH2Cl2 7. H2O + H2SO4 2. BH3/THF, followed by HO-, H2O2, H2O 5. Br2 + H2O 8. CH3OH + H2SO4 3. a peroxyacid 6. H2 + Pd/Carrow_forward1. Which reaction conditions would be best for turning ethene into ethanol? a.H2O, HBr b. CH3OH, H2SO4 c.CH3OH, HBR d. H2O, H2SO4 2. Which alkene would be non-regioselective in a reaction with HBr? a. 2-ehyl-2-methyl-2-butene b.2-ethyl-3-methyl-2-butene c. 2,3-dimethyl-2-butene d.1,2-dimethyl-2-butenearrow_forward
- 1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the abovearrow_forward1. What type of reaction is occuring in step 3? (halogenation, hydrohalogenation, reduction, keto–enol tautomerism, dehydrohalogenation, acid-catalyzed hydration, base-catalyzed hydration) 2. Which reagent is necessary for step 3? (Br2, HBr, H2/Pt, NaNH2, H20/H2SO4/HgSO4)arrow_forwardDraw the major organic product for the reaction between 1,2-dimethylcycloheptene and a cold, dilute solution of KMnO4.arrow_forward
- Name the structure. Which product is formed after the addition of Jones Reagent to compound B. (Compound B is the structure on the image)arrow_forward1.Label kinetic product 2.Label the thermodynamic product 3.Predict the four products from reaction of alkene with one equivalent HBrarrow_forwardWhat will be the major product of the reaction of 2-methyl-2-butene with each of the following reagents? a) Br2/H2O b)BH3/THF followed by H2O/Hydeoxyl ionarrow_forward

