
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 6.14, Problem 25P
Interpretation Introduction
Interpretation:
The enol tautomers for the given
Concept Introduction:
Conversion of terminal
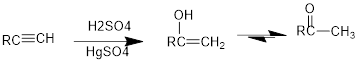
If a carbonyl group is bonded to two alkyl groups, it is called as a ketone. The enol formed in the acid catalysed addition of water will be easily converted into a ketone.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Identify the most stable enol tautomer of 3-methyl-1-phenyl-2-butanone.
Draw the product formed when the α,β-unsaturated ketone A is treatedwith following reagent.
[1] (CH2=CH)2CuLi; [2] H2O
Draw the major organic product obtained from the reaction of the following alkyl halide with benzoate.
Chapter 6 Solutions
Essential Organic Chemistry, Global Edition
Ch. 6.1 - Draw the mechanism for the reaction of cyclohexene...Ch. 6.2 - a. How many bond orbitals are available for...Ch. 6.2 - Prob. 3PCh. 6.2 - Prob. 4PCh. 6.3 - Prob. 5PCh. 6.3 - Prob. 6PCh. 6.3 - Prob. 7PCh. 6.5 - Prob. 9PCh. 6.5 - Prob. 10PCh. 6.5 - a. What is the major product of each of the...
Ch. 6.5 - Prob. 12PCh. 6.6 - What stereoisomers are obtained from each of the...Ch. 6.6 - Prob. 14PCh. 6.8 - Prob. 15PCh. 6.10 - Name the following:Ch. 6.10 - Draw the structure for each of the following: a....Ch. 6.10 - Draw the structures for and name the seven alkynes...Ch. 6.10 - Name the following:Ch. 6.10 - Name the following:Ch. 6.11 - What hybrid orbitals are used to form the...Ch. 6.13 - Prob. 22PCh. 6.14 - Prob. 23PCh. 6.14 - Which alkyne would be the best one to use for the...Ch. 6.14 - Prob. 25PCh. 6.14 - Prob. 26PCh. 6.15 - Describe the alkyne you would start with and the...Ch. 6.15 - What are products of the following reactions?Ch. 6 - Prob. 29PCh. 6 - Prob. 30PCh. 6 - Prob. 31PCh. 6 - Prob. 32PCh. 6 - What is each compounds systematic name?Ch. 6 - Prob. 34PCh. 6 - Prob. 35PCh. 6 - What reagents could be used to carry out the...Ch. 6 - Prob. 37PCh. 6 - Prob. 38PCh. 6 - Prob. 39PCh. 6 - Prob. 40PCh. 6 - Prob. 41PCh. 6 - Prob. 42PCh. 6 - Answer Problem 42 using 2-butyne as the starting...Ch. 6 - What is each compounds systematic name?Ch. 6 - Prob. 45PCh. 6 - Prob. 46PCh. 6 - Prob. 47PCh. 6 - Prob. 48PCh. 6 - Prob. 49PCh. 6 - Prob. 50PCh. 6 - Draw the keto tautomer for each of the following:Ch. 6 - Propose a mechanism for the following reaction...Ch. 6 - Prob. 53PCh. 6 - Prob. 54PCh. 6 - Prob. 55PCh. 6 - Propose a mechanism for the following reaction:Ch. 6 - Prob. 57PCh. 6 - Prob. 58PCh. 6 - Prob. 59PCh. 6 - Prob. 60PCh. 6 - Prob. 61PCh. 6 - Prob. 62P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the enol form of the following ketonearrow_forwardDraw the keto- tautomer for the following enolsarrow_forwardDraw the product formed when the α,β-unsaturated ketone A is treated with each reagent.a. NaBH4, CH3OH b. H2 (1 equiv), Pd-C c. H2 (excess), Pd-C d. [1] CH3Li; [2] H2O e. [1] CH3CH2MgBr; [2] H2O f. [1] (CH2=CH)2CuLi; [2] H2Oarrow_forward
- Draw the favored product(s) of the following reactions (organic chemistry)arrow_forwardWhich of the following pairs are keto–enol tautomers? a. CH3CH2CH=CHCH2OH and CH3CH2CH2CH2CH=O b. CH3CH-OHCH3 and CH3C=OCH3 c. CH3CH2CH=CHOH and CH3CH2CH2CH=O d. CH3CH2CH2CH=CHOH and CH3CH2CH2C=OCH3 e. CH3CH2CH2HO-C=CH2 and CH3CH2CH2C=OCH3arrow_forwardHow would you prepare the following compound using a Robinson annulation reaction between a β-diketone and an α, β-unsaturated ketone? Draw the structures of both reactants and the intermediate Michael addition product.arrow_forward
- Identify the reagents a-c in the following scheme:arrow_forwardDraw the most stable enol form for each of the following keto tautomersarrow_forwardRank the following esters in order of decreasing reactivity in the first slow step of a nucleophilic acyl substitution reaction (formation of the tetrahedral intermediate):Rank the same esters in order of decreasing reactivity in the second slow step of a nucleophilic acyl substitution reaction (collapse of the tetrahedral intermediate).arrow_forward
- Answer the following question about curcumin, a yellow pigmentisolated from turmeric, a tropical perennial in the ginger family and aprincipal ingredient in curry powder. Most enols, compounds that contain a hydroxy group bonded to a C=C, are unstable and tautomerize to carbonyl groups. Draw the keto form of the enol of curcumin, and explain why the enol is more stable than many other enols.arrow_forwardRank the following carbonyl compounds in order of increasing reactivity in nucleophilic substitution reactions:arrow_forwardDraw the enol or keto tautomer(s) of each compoundarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License