
Concept explainers
Interpretation:
The compound which will be more hydrated should be determined.
Concept introduction:
Carbocation: It is carbon ion that bears a positive charge on it.
Leaving group: It is a fragment that leaves from a substrate with a pair of electrons via
Carbocation stability order:
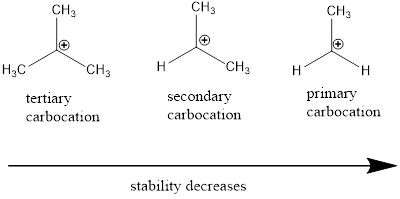
Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Essential Organic Chemistry, Global Edition
- The PKa of p-nitrophenol is 7.15. Would you expect this to dissolve in a sodium bicarbonate solution? The PKa of 2,5-dinitrophenol is 5.15. Will it dissolve in bicarbonate solution?arrow_forwardAcetal product formation is difficult, since hemiacetals are stable while acetals are not stable. True Falsearrow_forwardExplain why an acetal can be isolated but most hydrates cannot be isolated.arrow_forward
- Which of the following alcohol would react to Lucas reagent almost instantly?arrow_forward1. Which compound is expected to have a higher melting point, 1-pentanol or 1-pentanal? Explain 2. Which compound is expected to be more soluble in water, propanal or butanal? Explain. 3. Why is acetyl chloride more reactive towards hydrolysis than ethyl acetate?arrow_forwardI cyclohexanone II cyclohexanol III methylcyclohexanol. (b) Arrange the above compounds in the order of increasing boiling point. Explain. thank you for helping me.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

