
(a)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:

The equilibrium is favored to product side by a factor of
Explanation of Solution
The given reaction is:
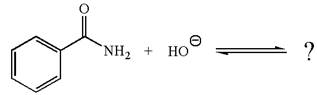
The ion

In the conjugate base formed, the negative charge on nitrogen is delocalized through the electron withdrawing resonance effect of carbonyl group. Thus, amide is a stronger acid than water, and hence, the equilibrium is favored to the product side.
The
The favored equilibrium side with numerical value is determined on the basis of stronger acid and
(b)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with the numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:
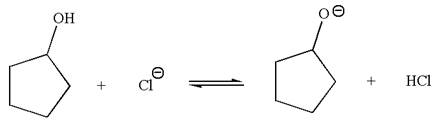
The equilibrium is favored to the reactant side by a factor of
Explanation of Solution
The given reaction is:
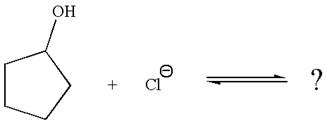
In the given reaction,
The favored equilibrium side with numerical value is determined on the basis of the stronger acid and
(c)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:
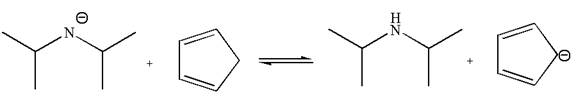
The equilibrium is favored to product side by a factor of
Explanation of Solution
The given reaction is:
In the given reaction, cyclopentadiene acts as an acid and the negatively charge nitrogen abstracts a proton from diisopropylamine to give the following products:
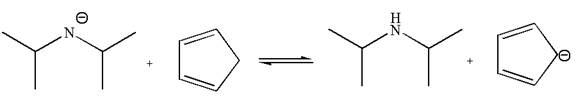
On the product side, the negative charge on carbon is a resonance stabilized by a conjugated double bond; such stabilization of the negative charge is not possible on the reactant side where the negative charge is on nitrogen bonded to two electron donating isopropyl groups. The acid is stronger when its conjugate base is stable, therefore, cyclopentadiene is a stronger acid than
According to Appendix
The favored equilibrium side with numerical value is determined on the basis of stronger acid and
(d)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:

The equilibrium is favored to the product side by a factor of
Explanation of Solution
The given reaction is:
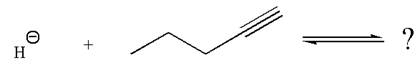
In the given reaction, the hydride ion abstracts the terminal proton of an

As the effective electronegativity of
According to Appendix
The favored equilibrium side with numerical value is determined on the basis of stronger acid and
(e)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:

The equilibrium is favored to the product side by a factor of
Explanation of Solution
The given reaction is:
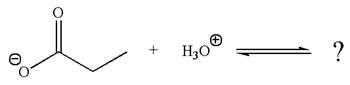
In the given reaction, the propanoate ion abstracts the proton of hydronium ion to give the following products:

According to Appendix
The favored equilibrium side with numerical value is determined on the basis of stronger acid and
(f)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:

The equilibrium is favored to the product side. Benzene is the weaker acid by a factor of
Explanation of Solution
The given reaction is:
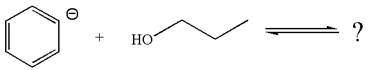
In the given reaction, the

As the oxygen atom is more electronegative than carbon, the negative charge on oxygen is more stable as compared to carbon. Thus, an anion on the right side, having negative charge on oxygen, is more stable than the anion on the left side where the negative charge is on carbon. Therefore, propanol is more acidic than benzene, and hence, the reaction is favored to the product side.
According to Appendix
The favored equilibrium side with numerical value is determined on the basis of stronger acid and the
(g)
Interpretation:
The products for the given proton transfer reaction are to be drawn and the favored equilibrium side with numerical factor is to be determined.
Concept introduction:
The proton transfer reactions favor the side opposite the stronger acid. Larger the
Answer to Problem 6.41P
The products for the given reaction are:

The equilibrium is favored to the product side. Benzene is the weaker acid by a factor of
Explanation of Solution
The given reaction is:
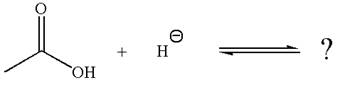
In the given reaction, the hydride ion abstracts the proton from carboxylic acid and gives the following products:

The conjugate base formed with a negative charge on the oxygen atom is better stabilized by the resonance effect. This makes the carboxylic acid the stronger acid, and the equilibrium is favored to the product side.
According to Appendix
The favored equilibrium side with numerical value is determined on the basis of stronger acid and the
Want to see more full solutions like this?
Chapter 6 Solutions
ORGANIC CHEMISTRY:PRIN...(PB)-W/ACCESS
- I understand where the proton transfer occurs but not what the products will look like. Can you draw curved arrows to show proton transfer and what the reaction products would look like? And explain which side is favored and whyarrow_forwardAt a ph below the pka of a weak acid there will be _ concentration of the deportonated form than the proton aged form Equal higher lowerarrow_forwardPlease explain in detail. What are the pka values you determined to predict that this molecule is more acidic?arrow_forward
- Could someone help me rank these in order of increasing activity in an SN1 reaction? Thanks!arrow_forwardWhich of the underlined protons for molecule 1 and 2 is more acidic and why?arrow_forwardStarting with [Pt(NH3)4]2+ or [PtCl4]2- and using the trans effect sequence, devise a rational procedure for synthesizing [Pt(py)(NH3)(NO2)Cl] with py and NH3 positioned trans to one another. Note, the fewer the steps, the better.arrow_forward
- The product in this reaction is basic enough to be protonated by a dilute HCl solution. Draw the protonated species, clearly showing where protonation occurs. Draw all possible resonance structures of the conjugate acid of the product, and use these to explain why the product is so much more basic than a typical ester, like ethyl acetate.arrow_forwardHello, Isn't there a conjugate acid/base for each of the structures?arrow_forwardThe answer key states this is an E2 reaction However I do not see how it could be with the Br being 3rd degree, KCN being weakly basic and DMF being polar aprotic. Would this not result in no reaction? I don't understand how this could turn into an E2 reaction.arrow_forward
- Please fill in the incomplete reactions , show the mechanism and state whether it is SN1, SN2, E1, E2 or proton transfer.arrow_forwardWhich of the compounds shown below is the most acidic hydrogen and what is the approximate pKa value?arrow_forwardHow do these mechanisms with NET3 work? Would someone be able to draw out the mechanisms? Specifically for the last one, I am confused because if NET3 is a base, shouldn't the double bond be the adjacent one (tetra substituted)?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
