
(a) Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanol as a solvent with respect to leveling effect.
Concept introduction:
The solvent affects the properties of bases and acids. This effect is referred as leveling effect. For an acid-base reaction, the basicity of the solvent levels or limits the strength of the strong acid. Similarly, the acidity of the solvent levels the strength of the strong base. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.45P
With respect to the leveling effect, ethanol is not a suitable solvent for a reaction involving
Explanation of Solution
The reaction of
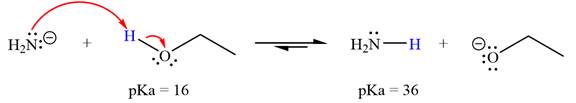
Ethanol,
The solvent effect on the reactant is determined with respect to the leveling effect.
(b)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanol as a solvent with respect to leveling effect.
Concept introduction:
The solvent affects the properties of bases and acids. This effect is referred as leveling effect. For an acid-base reaction, the basicity of the solvent levels or limits the strength of the strong acid. Similarly, the acidity of the solvent levels the strength of the strong base. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.45P
With respect to the leveling effect, ethanol is a suitable solvent for a reaction involving
Explanation of Solution
The reaction of the acetate ion
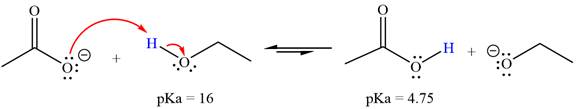
Acetic acid,
The solvent effect on the reactant is determined with respect to the leveling effect.
(c)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanol as a solvent with respect to leveling effect.
Concept introduction:
The solvent affects the properties of bases and acids. This effect is referred as leveling effect. For an acid-base reaction, the basicity of the solvent levels or limits the strength of the strong acid. Similarly, the acidity of the solvent levels the strength of the strong base. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.45P
With respect to the leveling effect, ethanol is a suitable solvent for a reaction involving chloride ion
Explanation of Solution
The reaction of chloride ion
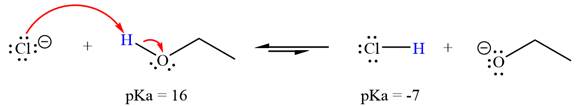
Hydrochloric acid,
The solvent effect on the reactant is determined with respect to the leveling effect.
(d)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanol as a solvent with respect to leveling effect.
Concept introduction:
The solvent affects the properties of bases and acids. This effect is referred as leveling effect. For an acid-base reaction, the basicity of the solvent levels or limits the strength of the strong acid. Similarly, the acidity of the solvent levels the strength of the strong base. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.45P
With respect to the leveling effect, ethanol is a suitable solvent for a reaction involving phenoxide ion (
Explanation of Solution
The reaction of phenoxide ion (
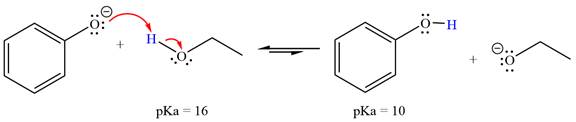
Phenol,
The solvent effect on the reactant is determined with respect to the leveling effect.
(e)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanol as a solvent with respect to leveling effect.
Concept introduction:
The solvent affects the properties of bases and acids. This effect is referred as leveling effect. For an acid-base reaction, the basicity of the solvent levels or limits the strength of the strong acid. Similarly, the acidity of the solvent levels the strength of the strong base. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.45P
With respect to the leveling effect, ethanol is a suitable solvent for a reaction involving
Explanation of Solution
The reaction of cyanide ion
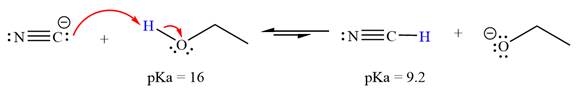
Acetylene,
The solvent effect on the reactant is determined with respect to the leveling effect.
(f)
Interpretation:
It is to be determined whether the given reactant is suitable for a reaction involving ethanol as a solvent with respect to leveling effect.
Concept introduction:
The solvent affects the properties of bases and acids. This effect is referred as leveling effect. For an acid-base reaction, the basicity of the solvent levels or limits the strength of the strong acid. Similarly, the acidity of the solvent levels the strength of the strong base. With respect to the leveling effect, a solvent is unsuitable for a particular reactant R if R is a stronger acid that the solvent’s conjugate acid (i.e., R has the lower pKa) or if R is a stronger base than the solvent’s conjugate base (i.e., the conjugate acid of R has a higher pKa than the solvent).
Answer to Problem 6.45P
With respect to the leveling effect, ethanol is not a suitable solvent for a reaction involving
Explanation of Solution
The reaction of propyl group
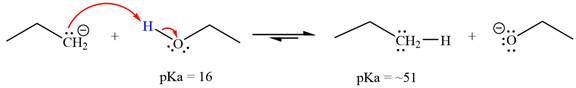
Ethanol,
The solvent effect on the reactant is determined with respect to the leveling effect.
Want to see more full solutions like this?
Chapter 6 Solutions
EBK GET READY FOR ORGANIC CHEMISTRY
- The addition of water to aldehydes and ketones occurs rapidly, although it is not thermodynamically favored. What would be the product for the reaction above? Hint: Think of the self-ionization of water and the polarity of the carbonyl group.arrow_forwardDon't use hand raiting pleasearrow_forwardTOPIC: Preparation of Ethylene Expound and explain the given answer to the question.arrow_forward
- 6. Describe concisely a chemical test to distinguish between the following pairs of compounds. Terangkan secara ringkas ujian kimia bagi membezakan pasangan sebatian berikut. (a) Propanal and propanone (b) Phenol and benzoic acid (c) Hexan-3-one and hexan-2-onearrow_forwardPlease help me with the following Organic Chemistry question. Please give explanation where necessary.arrow_forwardUse the following atomic weights and quantities to calculate the overall % yield of tetraphenylcyclopentadienone. Remember that your yield must be based on the limiting reagent, and that grams must be converted to moles. Filling out most of the chart will help. Give only two significant digits in your answer. If after rounding the answer is a whole number, do not include a decimal point. C = 12, H = 1, K = 39, O = 16 benzil + dibenzyl ketone potassium + → tetraphenylcyclopentadienone hydroxide formula formula weight grams 2.314 7.231 3.857 3.173 moles Answer: % yieldarrow_forward
- Use the following atomic weights and quantities to calculate the overall % yield of tetraphenylcyclopentadienone. Remember that your yield must be based on the limiting reagent, and that grams must be converted to moles. Filling out most of the chart will help. Give only two significant digits in your answer. If after rounding the answer is a whole number, do not include a decimal point. C = 12, H = 1, K = 39, O = 16 formula formula weight grams moles Answer: benzil + dibenzyl ketone + potassium hydroxide tetraphenylcyclopentadienone 4.135 8.646 1.140 % yield 6.956arrow_forwardWhich of the following is true regarding the Heck coupling reaction? 1)Bond formation can occur between an aryl halide and an alkene in the presence of a palladium-ligand catalyst. 2)Two carboxylic acid derivatives can be linked together in the presence of a palladium-ligand catalyst 3)An alkene and alcohol are linked together in the presence of a palladium-ligand catalyst. 4)Two alkenes can be linked together in the presence of a palladium-ligand catalyst.arrow_forwardNomenclature: provide the systematic names or structure of each of the following compounds. Include E/Z/R/S where relevant.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


