
Concept explainers
Interpretation:
The given questions under given set of conditions should be answered.
Concept introduction:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
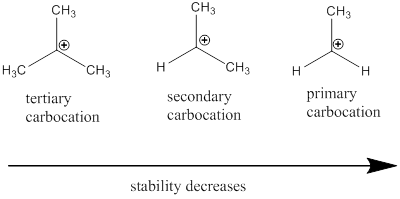
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
CHEM 262 ORG CHEM EBOOK DIGITAL DELIVERY
- Draw all resonance structures for the carbocation formed by ortho attack of the electrophile +NO2 on attached starting material. Label any resonance structures that are especially stable or unstablearrow_forwardDraw the structure(s) of the major organic product(s) of the following reaction. s ● PPh₂ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. Do not include lone pairs in your answer. They will not be considered in the grading. • Omit the phosphine oxide from your answer. . If no reaction occurs, draw the organic starting material. · • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the + sign from the drop-down menu. + 99-81 ***11 / / | ? (10 ChemDoodle Sn [Farrow_forwardAs we will learn in Section 13.12, many antioxidants–compounds that prevent unwanted radical oxidation reactions from occurring–are phenols, compounds that contain an OH group bonded directly to a benzene ring. a. Explain why homolysis of the O–H bond in phenol requires considerably less energy than homolysis of the O–H bond in ethanol (362 kJ/mol vs. 438 kJ/mol). b.Why is the C–O bond in phenol shorter than the C–O bond in ethanol?arrow_forward
- A benzene ring alters the reactivity of a neighboring group in the so-called “benzylic” position, similarly to how a double bond alters the reactivity of groups in the “allylic” position. Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates. a) Use resonance structures to show the delocalization of the positive charge, negative charge, and unpaired electron of the benzyl cation, anion, and radical.arrow_forward1. Which of the following cannot be a nucleophile? a. Ib. IIc. III2. Which of the following cannot be an electrophile? a. Ib. IIc. IVarrow_forward1c. What is the nucleophile in the following reaction? .Br CH;CO0 Na LO OCCH3 NaBr ld. What is the electrophile in the following reaction? Br CH;COO Na L0OCCH3 NaBr +arrow_forward
- How does gold help to catalyze the reaction in the gold catalysis experiment? It deprotonates the methanol, making methanol a better nucleophile. оа. o b. It donates electron density to the alkyne, making the alkyne a better nucleophile. О с. It coordinates to the methanol, making methanol a better nucleophile. o d. It coordinates to the alkyne, making the alkyne a better electrophile.arrow_forwardorgo chem questionsarrow_forwardProvide the whole reaction mechanism (generation of electrophile, nucleophile, bond formation, bond breaking and movement of arrows) and the final product of the following reactions:arrow_forward
- The two reactants shown below are combined to bring about a nucleophilic substitution reaction. [References] a + Nal C. Br e. I i. HI HỌ. g. HBr j. Na* k. Br h. H20 1. none Which letter designates the electrophilic carbon at which substitution occurs? (If no reaction occurs enter the letter corresponding to "none.") Which letter corresponds to the leaving group? Which letter designates the nucleophile?arrow_forwardDoes perchlorate (ClO4-) have an extremely low nucleophilicity because it participates in a lot of resonance structures?arrow_forwardI am confused about what the three categories are. Are they talking about lone pairs, pi bonds, and sigma bonds?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
