
Concept explainers
(a)
Interpretation:
The differences between the first step in reactions of
Concept introduction:
Addition Reaction: It is defined as
In addition reaction of
Regioselective reaction: They are reactions which contain more than one product which are actually molecules with same molecular formula but different in the way they are connected and among those products only one product is major.
Markovnikov’s Addition Rule: The unsymmetrical alkene in a chemical compound reacts with hydrogen halide in a way, where halide ions attacks and bond to the more substitution position of carbon-carbon double bond.
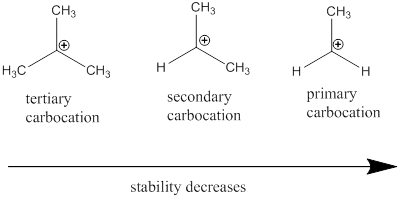
Carbocation: it is carbon ion that bears a positive charge on it.
Leaving group: it is a fragment that leaves from a substrate with a pair of electrons via
Carbocation stability order:
(b)
Interpretation:
The product and reason for attack of
Concept introduction:
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of alkenes when two substituents are placed on same side of
Regioselective reaction: They are reactions which contain more than one product which are actually molecules with same molecular formula but different in the way they are connected and among those products only one product is major.
Markovnikov’s Addition Rule: The unsymmetrical alkene in a chemical compound reacts with hydrogen halide in a way, where halide ions attacks and bond to the more substitution position of carbon-carbon double bond.
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry
- 1. Why is 5-octene not possible? 2. Why is 3,3-dimethyl-3-octene not possible?arrow_forwardWhat is the major product obtained from the reaction of propene and Br2 plus excess Cl-?arrow_forward2. In the bromination reactions, what is the function of CCl4? Why can it fulfil its role?3. Bromination proceeds by either free radical substitution or electrophillic addition. Based on Table 3, which mechanism is followed by alkanes? by alkenes? by alkynes?4. For which hydrocarbon type is light necessary for bromination to take place?arrow_forward
- complete the reactions draw the product from the reaction of HBr and 2-butenearrow_forwardDraw the structures of the two carbocation intermediates that might form during the reaction of propene (above) with HBr.arrow_forwardwhat is a less preferred way to make FAME'S rather than transesterification? why is is less preferred?arrow_forward
- When you react ammonia with a halogenated Alkane will you get only one organic product? Why or why notarrow_forwardwhich would be expected to convert 1 mole of 4-methyl-1-pentyne into 2-methylpentane? Na, NH3(l) 2 moles H2, Pt 1 mole H2, Pt 2 moles of HCl H2, Lindlar’s catalystarrow_forwardDoes Markovnikov's rule apply to the addition of HX to vinyl halides ?arrow_forward
- In the reaction of ethylene with H20O in the presence of sulfuric acid,which one adds across the double bond first? *arrow_forwardIn the reaction of ethylene with H20 in the presence of sulfuric acid,which one adds across the double bond first? * ulfate ionarrow_forwardMake propane from 2- bromo propane, show the synthetic pathwayarrow_forward

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT

