
(a)
INTREPRETATION:
The products obtained from addition of
CONCEPT INTRODUCTION:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
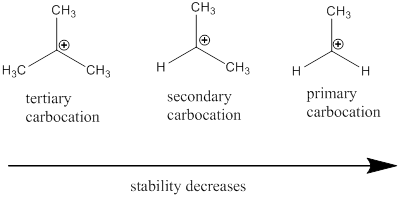
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:
(b)
INTREPRETATION:
The mechanism for addition of
CONCEPT INTRODUCTION:
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Chemical reaction involves bond making and breaking of two or more reactants in order to attain products from the reactants.
The curved arrows are generally used to indicate the flow of electrons present in the reaction.
Addition Reaction: It is defined as chemical reaction in which two given molecules combines and forms product. The types of addition reactions are electrophilic addition, nucleophilic addition, free radical additions and cycloadditions. Generally, compounds with carbon-hetero atom bonds favors addition reaction.
In addition reaction of alkenes when two substituents are placed on same side of
Oxidation Reaction: It involves loss of electrons, addition of oxygen atoms or removal of hydrogen atoms.
Oxidizing Reagents: The chemical agents used to add oxygen or remove hydrogen which finally reduced on oxidizing the other compound.
Carbocation: it is carbon ion that bears a positive charge on it.
Carbocation stability order:

Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry
- Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reactions below. You may omit NBS in your mechanism, and use Br and Br2.arrow_forwardPropose a mechanism for the following reaction: a. Is the initially formed carbocation primary, secondary, or tertiary? b. Is the rearranged carbocation primary, secondary, or tertiary? c. Why does the rearrangement occur?arrow_forwardDraw the SNl or SN2 reaction with CN for the following molecules t butylchloride , n - butylbromide , 2 -bromobutane . Explain in which mechanism it will react and why ?arrow_forward
- 20. (a) Show all steps in the synthesis of 2-pentene from 2-bromopentane. Show the structure of all reactants and product. CH3CHBrCH2CH2CH3 -------> CH3CH=CHCH2CH3 (b) Correctly identify the mechanism in (a) above(SN2, SN1, E2, E1)arrow_forwardDrawing Substitution and Elimination Products from a 2° Alkyl Halide. Explain how ?arrow_forwardThe following reaction takes place several times faster than the reaction of 2-chlorobutane with HO-: a. Explain the enhanced reaction rate.b. Explain why the OH group in the product is not bonded to the carbon that was bonded to the Cl group in the reactant.arrow_forward
- Reaction of 2° alcohol A with HCl forms three alkyl chlorides, all of which result from rearrangement of the 2° carbocation initially formed. Draw the structures of these products and a mechanism that illustrates how each is formed.arrow_forwardTwo elimination products are obtained from the following E2 reaction: a. What are the elimination products?b. Which is formed in greater yield?arrow_forwardTwo elimination products are obtained from the following E2 reaction: a.What are the elimination products? b. Which is formed in greater yield?arrow_forward
- 1. Which among these would be the most stable carbocation?a. Allylicb. Vinylicc. Primaryd. Secondary2. What is the primary reason for the stability of tertiary carbocations?a. Free rotationb. Resonancec. Hyperconjugationarrow_forward19. (a) Show the structure of all reactants and products for the bromination of 1,2 dimethylcyclopentene. Show the proper stereochemistry of the products. 1,2-dimethylcyclopentene + Br2/CCl4-----> Products (b) Show the mechanism of the reaction in (a) above.arrow_forwardConsider the E1 reaction of 3-bromo-2-cyclohexyl-butane. How many elimination products are possible? Include (E)/(Z) isomers as separate products.arrow_forward


 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


