
Concept explainers
Interpretation:
The compound which will be more hydrated should be determined.
Concept introduction:
Carbocation: It is carbon ion that bears a positive charge on it.
Leaving group: It is a fragment that leaves from a substrate with a pair of electrons via
Carbocation stability order:
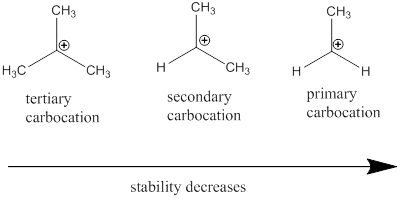
Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Nucleophile: Nucleophiles are electron rich compounds which donates electrons to electrophilic compounds which results in bond formation.
Nucleophilic nature depends on the negative charge present in the molecule, the solvent in which it present and the electronegativity of the atom.
Electrophile: Electrophiles are electron deficient compounds which accepts electrons from nucleophiles that results in bond formation.
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
Organic Chemistry
- What starting material is needed to prepare each compound by analdol reaction?arrow_forwardWhich ester hydrolyzes more rapidly?a. methyl acetate or phenyl acetate? b. phenyl acetate or benzyl acetate?arrow_forwardWhat starting materials are needed to prepare each compound using a Heckreaction?arrow_forward
- Draw the product formed from a Michael reaction with the given starting materials using −OEt, EtOH.arrow_forwardDraw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs. [1] C6H5Li (excess); [2] H2Oarrow_forwardWhich of the following compounds reacts most rapidly in a nitration reaction?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

