
Concept explainers
(a)
Interpretation:
An oxygen-binding curve for a hypothetical two subunit hemoglobin with
Concept introduction:
Hill equation is represented as follows:
Here, Y- fractional saturation
n − a measure of the degree of cooperativity in ligand binding.
P50 − partial pressure of oxygen at which hemoglobin is half saturated.
The Hill plot of log(Y/1-Y) versus log(P50) should be a linear graph with slope of n.
Answer to Problem 14P
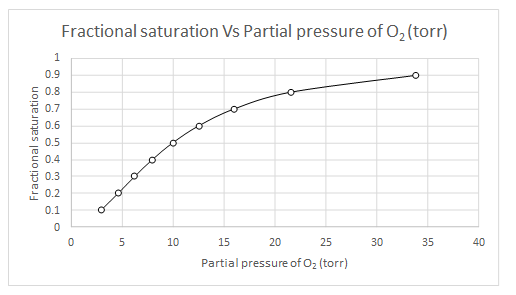
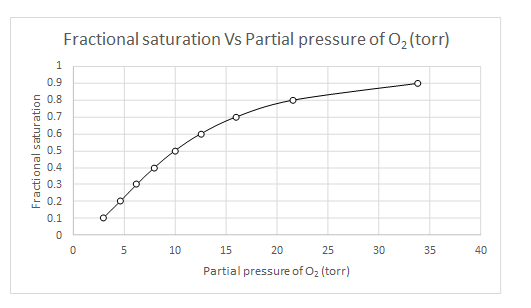
Explanation of Solution
The values of
| Y | log(Y/(1-Y) | log(Y/(1-Y)-n log (P50) | log (pO2) | pO2 |
| 0.1 | -0.95424251 | 0.845757491 | 0.469865 | 2.950294 |
| 0.2 | -0.60205999 | 1.197940009 | 0.665522 | 4.629374 |
| 0.3 | -0.36797679 | 1.432023215 | 0.795568 | 6.245518 |
| 0.4 | -0.17609126 | 1.623908741 | 0.902172 | 7.983099 |
| 0.5 | 0 | 1.8 | 1 | 10 |
| 0.6 | 0.176091259 | 1.976091259 | 1.097828 | 12.52646 |
| 0.7 | 0.367976785 | 2.167976785 | 1.204432 | 16.01148 |
| 0.8 | 0.602059991 | 2.402059991 | 1.334478 | 21.60119 |
| 0.9 | 0.954242509 | 2.754242509 | 1.530135 | 33.89493 |
(b)
Interpretation:
An oxygen-binding curve for shypothetical two subunit hemoglobin with
Concept introduction:
Concerted model equation is written as follows:
Here, Y − fractional saturation
a − ratio between the substrate concentration and the dissociation constant for a ligand binding to a single site in R state.
L − The ratio of the concentrations of the T and R states with no ligands bound
c − the ratio between the dissociation constant for a ligand binding to a single site in R state and that of T state.
n − number of binding sites
Also, the ratio can be calculated as follows:
Here,
pO2 − partial pressure of oxygen
KR - the dissociation constant for a ligand binding to a single site in R state.
Answer to Problem 14P
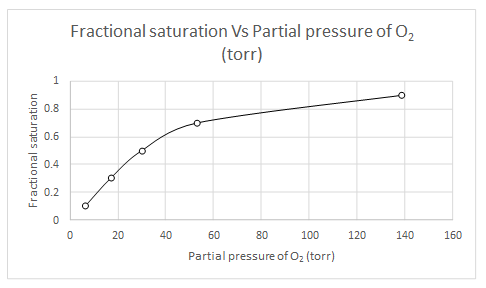
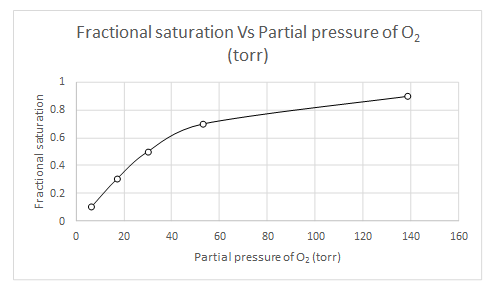
Explanation of Solution
When
Likewise, partial pressure of oxygen for several fractional saturation values are calculated and a plot of fractional saturation versus oxygen partial pressure is drawn.
| Y | pO2 |
| 0.1 | 6.55 |
| 0.3 | 17.09 |
| 0.5 | 30.15 |
| 0.7 | 53.21 |
| 0.9 | 138.91 |
Want to see more full solutions like this?
Chapter 7 Solutions
BIOCHEMISTRY 2 TERM ACCESS
- 384 Structure and Function of Hemoglobin Q4.4- In addition to O2, another diatomic molecule that can bind to the iron atom in hemoglobin is CO. Explain in biochemical terms why high levels of CO gas are lethal and cite a specific example of accidental death by CO poisoning (where/when/how).arrow_forward"Hemoglobin can be in either an oxy- or deoxy- state at a given pH. Given the appropriate pKa values, calculate fraction protonated at a given pH" How would I do this?arrow_forward(No more than one page). Explain why quaternary structure is necessary for cooperativity and allosteric regulation. Use hemoglobin as an example. Include the various allosteric effectors that influence oxygen binding.arrow_forward
- Oxygen-hemoglobin curve dissociation curve: Which various factors can determine the amount of saturation? why is this curve so steep? how ph and temperature affect it? how does the binding of oxygen molecules in bohr and haldane effect affect the 3d shape of hemoglobin and how does that have to do with the curve?arrow_forwardComparison of Fetal and Maternal Hemoglobins.Studies of oxygen transport in pregnant mammals show that the O2-saturation curves of fetal and maternal blood are markedly different when measured under the same conditions. Fetal erythrocytes contain a structural variant of hemoglobin, HbF, consisting oftwo a and two g subunits (α2γ2), whereas maternal erythrocytes contain HbA (α2β2). a)Which hemoglobin has a higher affinity for oxygen under physiological conditions, HbA or HbF? Explain. b)What is the physiological significance of the different O2 affinities? c)When all the BPG is carefully removed from samples of HbA and HbF, the measured O2-saturation curves (and consequently the O2affinities) are displaced to the left. However, HbA now has a greater affinity for oxygen than does HbF. When BPG is reintroduced, the O2-saturation curves return to normal, as shown in the graph. What is the effect of BPG on the O2affinity of hemoglobin? How can the above information be used…arrow_forwardQuestion- Some of the reactions of NO in the blood do not cause problems at low concentrations but can upset the normal reactions of hemoglobin if there is a large concentration of NO. The same reactions can cause trouble if a synthetic blood substitute contains a molecule similar to hemoglobin but does not contain all the other enzymes normally contained in red blood cells. Explain what this problem is and how it arises.arrow_forward
- Need help, please. Draw an oxygen binding curve of Hb at a pH of 7.4 and another curve where all 2,3 BPG has been removed Draw an oxygen binding curve of Hb at a pH of 7.4 and another at pH. Draw an oxygen binding curve of Hb at a pH of 7.4 and another with a mutant Hb in which the predominant form of the protein is monomericarrow_forwardIn a molecular disease of hemoglobin, Hemoglobin Rainier, Tyr 145β is replaced by Cys, which forms a disulfide bond with another Cysresidue in the same subunit. This prevents the formation of ion pairs that normally stabilize the T state. How does hemoglobin Rainier differ from normal hemoglobin with respect to (A)oxygen affinity, (B)the Bohr effect, and (C)the Hillcoefficient? Explain your answers.arrow_forward6. Order: 40,000 units heparin in 1L of 0.9% NaCl to infuse at 25 mL/h. Calculate the hourly heparin dosage in mL/hour.?arrow_forward
- 30. The cooperative binding behavior of hemoglobin for oxygen is best explained by... Group of answer choices The movement of the proximal histidine upon O2 binding causes a structural change at the binding interface between subunits The oxidation of Fe2+ to Fe3+ and formation of the superoxide ion causes distortion of the protoporphyrin ring, altering binding interface between subunits The tetrameric nature of hemoglobin's quaternary structure directly confers cooperative binding activity The movement of the distal histidine upon O2 binding causes a structural change at the binding interface between subunits The binding of O2 causes a pH shift that changes the protonation state of amino acids located at the interface between subunits The solubility of hemoglobin in aqueous solution and its insolubility in nonpolar environmentsarrow_forwardPrevious Assignment: The figure below shows the bonding of the cytosine and guanine molecules. The O - H and H - N distances are each 0.110 nm. In this case, assume that the bonding is due only to the forces along the O - H - O, N - H - N and O - H - N combinations, and assume also that these three combinations are parallel to each other. Calculate the net force that cytosine exerts on guanine due to the preceding three combinations. Is this force attractive or repulsive? ---------------------------------------------------------------------------------- Electric Potential Energy: Base Pairing in DNA, IICalculate the electric potential energy of the guanine–cytosine bond, using the same combinations of molecules (O - H - O, N - H - N and O - H - N) as in the previous assignment (pls refer above)arrow_forwardRECALL Which of the following does not play a role in respiratory complexes: cytochromes, flavoproteins, iron-sulfur proteins, or coenzyme Q?arrow_forward
 BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning

